Reverse Osmosis Pretreatment: Removal of Iron in Groundwater Desalination Plant in Shupramant-Giza - A Case Study
Al-Sayed M. Aly1 , Mahmoud M. Kamel2 , A. Hamdy1 * , Khaled Z. Mohammed1 and Mohamed A. Abbas1
1
Egyptian Petroleum Research Institute (EPRI),
Nasr City,
Cairo
Egypt
2
Chemistry Department,
Al-Azhar University (Assuit Branch),
Egypt
DOI: http://dx.doi.org/10.12944/CWE.7.1.04
Reverse osmosis (RO) is being increasingly utilized throughout the world for desalination due to the latest improvements in RO membrane performance and its reduced cost compared to thermal desalination. In this paper, Different media and chemicals have been used for Iron removal to prevent membrane fouling of groundwater reverse osmosis plant located in Shupramant-Giza. The objective is to present field results of the reverse osmosis plant operation in order to evaluate the reliability of this technology. The operating pressure and pressure drop increased significantly without an increase in the production capacity. Frequent shutdowns of the plant were observed due to severe membrane fouling. The membrane was cleaned with different chemical solutions to dissolve the deposits from the membrane surface. To achieve high cleaning efficiency, the flow rate of desalinated water and total dissolved salts (TDS) were studied.
Copy the following to cite this article:
Al-sayed MA, Mahmoud MK, Hamdy A, Khaled ZM, Abbas MA. Reverse Osmosis Pretreatment: Removal of Iron in Groundwater Desalination Plant in Shupramant-Giza - A Case Study. Curr World Environ 2012;7(1):23-32 DOI:http://dx.doi.org/10.12944/CWE.7.1.04
Copy the following to cite this URL:
Al-sayed MA, Mahmoud MK, Hamdy A, Khaled ZM, Abbas MA. Reverse Osmosis Pretreatment: Removal of Iron in Groundwater Desalination Plant in Shupramant-Giza - A Case Study. Curr World Environ 2012;7(1):23-32. Available from: http://www.cwejournal.org/?p=1738
Download article (pdf) Citation Manager Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2012-03-18 |
|---|---|
| Accepted: | 2012-05-20 |
Introduction
Due to recent developments in membrane technology, the trend in the desalination industry is to use reverse osmosis (RO) for desalting seawater. Brackish water (BW) desalination using membrane technology is also expanding as the salinity of groundwater increases. Selecting an appropriate process to meet specific needs at specific locations is essential though the biggest challenge remains in the capability to successfully operate these plants once installed due to peculiarities of sea and brackish waters in the region.1 Membrane filtration in general and reverse osmosis (RO) in particular is applied in a wide range of fields, such as chemical, medical, textile, petrochemical, electrochemical, water treatment, biotechnology and environmental industries.2
Fouling and scaling are the most serious problems in membrane processes. In sea/brackish water applications, pretreatment of RO feed water is the key step in designing the plants to avoid membrane fouling and scaling.1 At the present time, pretreatment technology is divided into conventional pre-treatment and non-conventional pre-treatment. Conventional RO pre-treatment has been widely applied for sea and ground water RO plants to achieve the expected quality of feed water to the RO membrane. But with the deterioration of feed waters and the consideration of the less efficient conventional system, an increasing number of plant owners were considering the use of membrane based pretreatments.3
Iron is found in surface and ground waters at varying concentration levels, usually up to 3–4 mg/l and in some cases up to 15 mg/l.4 Sharma ; et al., found that when present, even at low concentrations it can be linked to aesthetic and operational problems such as bad taste and color, staining, as well as deposition in the water distribution system leading to incidence of high turbidity.5 Also, iron promotes the growth of certain types of chlorine-tolerant microorganisms in water distribution systems, thus causing increased costs for cleaning and sterilizing systems in addition to odor and taste problems. The highest permitted limit of iron concentration for drinking water is 0.2 mg/l.6
Chemical cleaning of membrane means removing impurities by means of chemical agents. The first step of chemical washing is finding appropriate materials as cleaning agents. This depends on feed composition and precipitated layers on the membrane surface and in most cases is performed using a trial and error method.7 The cleaning agents must be able to dissolve most of the precipitated materials and remove them from the surface of membrane with no surface damage.8
The FilmTec Corporation was established in 1977 with the introduction of the FILMTEC FT30 reverse osmosis membranes which was the first commercially viable thin-film composite polyamide membrane for brackish water treatment. The FilmTec Corporation was purchased by the Dow chemical company in 1985, a move that merged Dow’s sales and marketing strength and expertise in polymer and membrane research with FilmTec’s membrane research, manufacturing and technical service resources.9 This paper includes evaluation to compare performance results during operation and operating cost of conventional media filtration, which is one of the most important decision-making bases for choosing feasible pretreatment methods.
Raw Water Characteristics
The raw water coming from two wells contains ca. 2 g/l total dissolved solids, predominantly chloride and sodium ions. The increase in the salinity represents only dissolved salts. Iron and manganese often occur together in groundwater but manganese usually occurs in much lower concentration than iron. Both iron and manganese are readily apparent in drinking water supplies. The highest permitted limit of iron concentration for drinking water is 0.2 mg/l.6 The feed water temperature is almost ranged in all seasons between 20 and 42°C. Raw water analysis by an Atomic Absorption Spectrometer (Perkine Elmer Flame AAS 3110) is presented in Table1.
RO Plant Characteristics
The feed water is supplied with two feed pumps with a specification: stainless steel 304, 20 m3/h – 5 bar max, kw 5.5, IP 55, class F. Feed water pumps are followed by dual media filter vessel. This vessel constructed of a fiberglass reinforced polyester resin for standard water conditioning use with specific size (diameter 13 inches (330 mm) and height 54 inches (1372 mm)), maximum operating pressure 150psi (10.34 bars), maximum operating temperature 120o F (48o C), bed capacity in liters is 105 and the top opening of this vessel is 2½ inches. Dual media filter vessel has two layers of filtration media – typical design includes anthracite,10 with effective size: 0.6-0.8 mm, sand11, 0.45-0.55 mm, and/or gravel, 2.0-3.0 mm, Table 2.
The vessel which used as media filter is controlled by automatic head conditioning controller that is a simple mechanical design, two valve body designs, one for downflow regeneration and one for upflow. Head controller has a choice of 7 or 12-day time clock or demand regeneration with either mechanical or electronic meter. The continues flow rate up to 20 gpm with regeneration time available up to 120 minutes and mounting base 2½ inches.
The high pressure pump with a specification: stainless steel 304, 20 m3/h – 17 bar max, k w15, IP 55, HP 20 and class F, supplies the pretreated water to the three membrane pressure vessels (housings) of the RO plant. Each housing contains one spiral wound polyamide membranes (Filmtec BW30-4040), Table 3. The membrane nominal active surface area is 7.6 m2; its permeate flow rate is 9.1 m3/d and the minimum salt rejection is 99.5%. Two flow meters are present to measure the in-and-out water of RO plant. Finally, the RO plant was controlled by electrical control panel.
Pretreatment Methods
Granular Media Filtration
Direct filtration, using mono, dual-media or mixed-media filtration, is the most common technology used for the filtration of seawater prior to the RO system. Filtration depends primarily on a combination of complex physical and chemical mechanisms, the most important being adsorption. As water passes through the filter bed, the suspended particles contact and adsorb (stick) onto the surface of the individual media grains or onto previously deposited material.13 To reach the expected quality of filtrate, the size, surface charge, and geometry of both suspended solids and filter media are the most important parameters that need to be well designed.
Water Desalination Technical Manual (WDTM), Department of the U.S. Army,14 gave the following design parameters for single, dual and mixed-media filtration:
1. Single-media filtration. Single-media filtration consists of one media. This media is often small-grained silica sand; however, anthracite may be used after lime and lime-soda softening. Some desalination pretreatment systems use an alternate media such as greensand to remove iron and manganese compounds. Diatomaceous earth media is not recommended for primary filtration because of its characteristic high head loss and short run times.
2. Dual media filtration. Dual media filtration consists of two media with different specific gravities. The difference creates a two-layer separation effect: The use of silica sand or greensand for one layer; or the use of anthracite for the other layer. The use of dual media will allow larger quantities of material to be filtered and will reduce head loss during operation. The use of two media types will provide a good coarse-to-fine filtration process for desalination facilities.
3. Mixed-media filtration. When three media are used in filters, a better coarse-to-fine filtration pattern can be obtained. High density silica sand, garnet, and anthracite are commonly used to provide the filter bed. The different media do not stratify completely. Instead, there is a small amount of intermixing among the different layers. This gradual change in media size provides a gradient from coarse to fine and creates a media flow pattern necessary to achieve a very low silt density index.
In this case, Dual media filter have two layers of filtration media – typical design includes anthracite, sand and/or gravel, Table3. The depth of the filter bed is typically a function of the media size and follows the general rule-of-thumb that the ratio between the depth of the filter bed (l - in millimeters) and the effective size of the filter media (de - in millimeters), l/de, should be higher than 1500. For example, if the effective size of the anthracite media is selected to be 0.6 to 0.8 mm, the depth of the anthracite bed should be at least (0.6 mm × 1500= 900 mm to 0.8 mm × 1500= 1200 mm, i.e., 0.9-1.2 m).15
In comparison to single sand filter media, dual filter media with anthracite over sand permit more penetration of the suspended matter into the filter bed, thus resulting in more efficient filtration and longer runs between cleaning. Periodically, when the differential pressure increase between the inlet and outlet of the pressure filter is 0.3–0.6 bar, and about 1.4 m for the gravity filter, the filter is backwashed and rinsed to carry away the deposited matter. Backwash time is normally about 10-120 min. Before a backwashed filter is placed back into service, it must be rinsed to drain until the filtrate meets the specification.16
Last, to protect the RO membrane from the breakthrough particles from media filtration, cartridge filters are usually recommended in the last step of a pre-treatment sequence. The pore size from 1 to 20 µm can be used based on different produced water quality from media filtration.14 In this case, we used cartridge filter with pore size 5 µm and length 20 inches. After filtration through these filters, the turbidity reduced from 3.87 NTU to 0.24 NTU.
Scale Inhibition
Scale inhibitors (antiscalants) can be used to prevent or control scaling. There are generally three different types of scale inhibitors: sodium hexa-metaphosphate (SHMP), organophosphonates and polyacrylates. According to FILMTEC Reverse Osmosis Membranes Technical Manual,17 SHMP is inexpensive but unstable compared to polymeric organic scale inhibitors. Hydrolysis of SHMP will not only decrease the scale inhibition efficiency, but also create a calcium phosphate scaling risk. Therefore, SHMP is generally not recommended. Organo-phosphonates are more effective and stable than SHMP. They act as anti- foulants for insoluble iron, keeping them in solution. Polyacrylates (high molecular weight) are generally known for reducing silica scale formation via a dispersion mechanism. Dosage rates on all antiscalants should be based on the antiscalant manufacturers. Overdosing should be avoided to make certain that no significant amounts of cationic polymers are present when adding an anionic scale inhibitor.18
In this case study, injection of antiscalant has done by chemical dosing pump (5 liters/7bars). Feed water pH was reduced from 6.78 to 6.52 by the effect of Permatreat 510 antiscalant which is a mixture of polymers and phosphonates. This antiscalant is specifically developed for groundwater with a high content of silica, and it is also effective with respect to precipitation of calcium salts (carbonate, sulfate and fluoride) and the fouling of iron (iron reduced from 3.8 mg/l to 3.12 mg/l).
pH Adjustment
Acidity (pH) adjustment is an efficient way to control scaling. By adding H+ as acid, the equilibrium can be shifted to keep salts dissolved. Adjustment chemicals to the pH include carbon dioxide, sulfuric acid, and hydrochloric acid. Carbon dioxide should not be used for pH adjustment of lime addition systems due to scaling problem associated with lime pretreatment. Sulfuric acid is easier to handle and in many countries more readily available than hydrochloric acid; however, additional sulfate is added to the feed stream, potentially causing sulfate scaling.13
In this case, it should be known that the pH is always changed significantly and the pH must be returned to a neutral state for the final produced water. At the beginning of the study, sulfuric acid is used. However, membrane fouling was observed. In order to stopping this fouling, the acid was then switched to hydrochloric for the remainder of the study. After the switch from sulfuric to hydrochloric acid, the plant worked very well, and the fouling is not observed according to standard permeate flow rate (27.3 m3/d) and TDS (50 mg/l).
Iron Removal Strategies
Iron, usually presents in groundwater as divalent ion (Fe2+) and is considered as source of membrane scaling. The main target in our case study is the removal of iron in groundwater before passing through reverse osmosis membranes as pretreatment technique to avoid membrane fouling. Take in account that the antiscalant feeding before membranes is effective with respect to precipitation. It reduces iron concentration from 3.8 mg/l to 3.12 mg/l, but this iron level is still the main source of membrane problems.
In this case, various treatment methods have been employed to enhance water quality by removing iron.
Oxidation Processes
Alternative processes have been proposed in order to facilitate the operation and to allow the removal of high amounts of iron in the presence, or absence, of dissolved organic matter. In both cases, a pH adjustment is necessary to maintain iron in the dissolved state to avoid membrane fouling.Ferrous iron is oxidized in air according to the following reaction:
Fe2+ + (1/4) O2+ H+ ”! Fe3+ + (1/2) H2 O ...(1)
Potassium Permanganate and Depth Filtration
Conventional treatment for iron removal from groundwater consists of oxidation and depth filtration. Oxygen or stronger oxidants, such as potassium permanganate (KMnO4), are generally used for Fe 2+ oxidation. The solid products of oxidation (FeOOH.H2O) are then filtered through a granular bed, commonly green sand.19 The potassium permanganate dose applied must be carefully controlled to minimize any excess passing into supply which could give a pink color to the water. Potassium permanganate oxidation tends to form a colloidal precipitates which may not be well retained by the filters.
Chlorine and Depth Filtration
The removal of iron along with chlorination step and appropriate dose of chlorine will be discussed. In particular membrane fouling caused by oxidized particles, was assessed in depth with visualization of the membrane surfaces. As shown in Fig.1, the removal efficiency of dissolved iron increased very rapidly and reached nearly 100% within 20 minutes with the appropriate dose of chlorine, 2.75 mg/L. With a higher dosage of chlorine 2.75 mg/L, there was no significant increase in the removal of metal ions but more serious membrane fouling occurred. The use of chlorine may be inadvisable when treating waters containing organic substances due to the possibility of disinfection byproducts (DBPs) formation.
Manganese Greensand
An alternative filter media is manganese green sand,20 formed by treating greensand (glauconite), which is a sodium zeolite, with manganous sulphate followed by potassium permanganate. Mn-greensand removes soluble iron by a process of ion exchange, frequently with the release of hydrogen ions. The process is therefore pH dependent, being virtually ineffective below pH 6.0 and very rapid at pH values above 7.5. When the Mn-greensand is saturated it is regenerated by soaking the filter bed with weak potassium permanganate solution. This procedure oxidizes iron on the surface of Mn-greensand thereby reactivating the exchange sites. It is reported that the exchange capacity is 1.45 g of Fe /l of Mn-greensand and that 2.9 g of potassium permanganate (as a 1% w/v solution) per liter of Mn-greensand is required for regeneration21. Alternatively, potassium permanganate is continuously applied to the bed by dosing it at the filter inlet, which maintains Mn-greensand active and catalyses the oxidation reaction. Mn-greensand then acts as a filter medium in addition to catalytic oxidation of any residual soluble manganese and is usually capped with a layer of anthracite to achieve longer filter runs. Operating the bed after oxidation capacity is exhausted will reduce its service life and may cause stain.
Oxidation and Microfiltration
This treatment is similar to the conventional one except that depth filtration is replaced by microfiltration (MF). The expected advantage of this treatment is to have a compact separation unit which produces high quality water from a wide range of raw water quality. In the present study the MF of iron oxide suspensions is removed.22,23
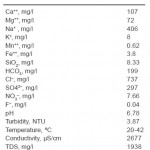 |
Table 1: Groundwater Composition Click here to View table |
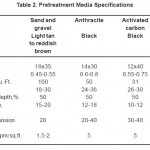 |
Table 2: Pretreatment Media Specifications Click here to View table |
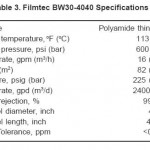 |
Table 3: Filmtec BW30-4040 Specifications. Click here to View table |
 |
Table 4: Amberlite IR120Na Data Sheet Click here to View table |
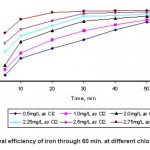 |
Figure 1: Removal efficiency of iron through 60 min. at different chlorine dosages. Click here to View table |
Finally, under certain conditions, the presence of free chlorine and other oxidizing agents, in the oxidation processes, will cause premature membrane failure. Since oxidation damage is not covered under warranty, FilmTec recommends removing residual free chlorine and other oxidizing agents by another suitable pretreatment prior to membrane exposure.24
Ion Exchange Resin
Ion exchange resins are able to remove many inorganic metal ions from groundwater including iron. In this case, Amberlite IR120Na, strong acid cation exchanger was used, Table 4.
Ion exchanger was carried out in a vessel constructed of a fiberglass reinforced vinylester resin for standard water de-ionizing use with specific size (diameter 13 inches and height 54 inches), maximum operating pressure 150psi (10.34 bars), maximum operating temperature 150o F (66oC), bed capacity in liters is 105 and the top opening of this vessel is 2½ inches. The total hardness concentration averaging 528 mg/L was passed through sodium charged strong acid cation exchange resin to reduce the hardness to less than 5 mg/L. Amberlite IR120Na, also treat with other metal ions like iron and so, the total exchange capacity is become smaller. The resin was then regenerated using commercially available extra coarse water-softening salt (NaCl). This process was repeated several times to demonstrate that no irreversible fouling had occurred to resin.
Granular Activated Carbon
Activated carbon26 is prepared from a char form material such as almond, coconut, and walnut hulls, other woods, and coal. Activated carbon has the strongest physical adsorption forces or the highest volume of adsorbing porosity of any material known to mankind. It is a highly porous material; therefore, it has an extremely high surface area for contaminant adsorption.27 The objective of this topic was to determine the effectiveness of granular activated carbon (GAC) in removing iron from the groundwater. From these advantages for granular activated carbon, in this case study, we used a single-media filter, Table 3. The depth of the GAC media is estimated based on the average contact time in this media, which is recommended to be 10 to 12 min. For example, if a filter is designed for a surface loading rate of 4 m3/m2 h, the depth of the GAC media should be at least 0.66 m (4 m3/m2 h ×10 min/60 min per h=0.66 m to 4 m3/m2 h ×12 min/60 min per h=0.8 m, i.e., 0.66 0.8 m).15 For the following reasons,28 we used the granular activated carbon in the adsorption of ferrous.
The van der Waals force that forms multilayer adsorption was overcome by the adsorbate due to the high ambient temperature.29 With relatively high room temperature of about 30oC where the adsorption process occurs, the chemisorption was more dominant as compared to the physisorption. The relatively high room temperature cause the chemical bond to occurs between the metal ions. Furthermore desorption will also occur between adsorbate and activated carbon at high temperature which physically bonded by the van der Waals force. Adsorbates which are physically adsorbed onto activated carbon receive sufficient energy from such high temperature to overcome the van der Waals force.
Activated carbon has high adsorption capacity for Fe(II) as compared to others. This may relate to adsorbate characteristics in terms of electronegativity. The electronegativity of Fe(II) is 1.8 . In fact, electronegativity is a measure of strength for element to attract electron. In this case, it would measure the strength of Fe(II) attach to negative charge at activated carbon surface. According to previous literature,30 higher electronegativities corresponded to the higher adsorption levels of metal ions onto the GAC.
Another factor that contributes to different GAC adsorption capacity on metal ion is ionic radius. Fe(II) has relatively smaller ionic radius than that of the others since Fe(II) has the higher attractive charge in nucleus on the electron orbital.29 The smaller ionic radius of Fe(II) makes it easier to penetrate into the micropores of the GAC.
There were four major functional groups on the surface of activated carbon which are carboxyl, carbonyl, hydroxyl, and lactonized carboxyl.31 All these four functional groups were promoted to attract cation to it and ion exchange would occur. Therefore, the Fe(II) which has positive charge would react and attach onto GAC surface’s functional groups with chemically bonded. However, the actual chemical reaction between the metal ion and functional groups on the activated carbon surface was complex and difficult to understand.
In the case of iron, oxidation is followed by settling and filtration or filtration alone, depending on the concentration of iron in the water. In the presence of turbidity (and color) and when the Fe(II) concentration is greater than about 5 mg/ l, settling or flotation would be assisted by a coagulant and/or a coagulant aid. Direct filtration is used when the iron concentration is less than about 5 mg/l.32
Post-Treatment Strategies
Post-treatment1 is limited to injection of lime to increase the pH from 6.52 to 8.0 and chlorine for disinfection.
Lime Post-Treatment
Lime has been added to neutralize the final produced water. For excess lime injection, it is necessary to raise pH to approximately 8. The high pH level produces good disinfection as a by-product and thus chlorination might be unnecessary after such injection except for a small dose to provide residual chlorine in the distribution system. Carbonation is necessary to remove the excess lime and reduce the pH after treatment.
Disinfection
Groundwater may be contains microorganisms such as bacteria, algae, fungi, and viruses, which can cause serious biological fouling. There are various methods to prevent and control biological fouling such as the addition of chemical oxidants (chlorine, bromine, iodine, or ozone), ultraviolet irradiation, biofiltration to remove nutrients, and the addition of biocide. Because of the risk of oxidation of the membrane, the use of oxidants must be monitored carefully to keep the chlorine well below 0.1 mg/L of free chlorine residual. Sometime dechlorination upstream of the membranes is required through sulfite compound addition or passage through granular-activated carbon.18
World Health Organization (WHO)33 considers: ‘it has been demonstrated that virus-free water can be obtained from faecally polluted source waters’ if the following chlorine disinfection conditions are met.
The water has a turbidity of 1 Nephelometric turbidity unit ( NTU) or less, Its pH is below 8.0, A contact period of at least 30 minutes is given; and, The chlorine dose applied is sufficient to achieve at least 0.5 mg/l free residual chlorine during the whole contact period.
Sodium hypochlorite (NaOCl) was used in our case study and the injection of hypochlorite has done by chemical dosing pump (5 liters/7bars) in dosage 1 mg/l. Typically iron should be less than 0.2 mg/l. If at the point of chlorine application, their levels are too low to justify disinfection, the dose must take their demand into account.
Membrane Cleaning
The fouling of RO elements is unavoidable with long-term operation. They can be fouled by biological matter, colloidal particles, mineral scale, and insoluble organic constituents. Deposits build up on the membrane surfaces during operation until they are causing loss in normalized permeate flow (product flow rate) and/or loss of normalized salt rejection [total dissolved salts (TDS)]. Elements should be cleaned whenever the normalized permeate flow drops by ≥10%, or the normalized salt passage increases by ≥10%, or the normalized differential pressure (feed pressure minus concentrate pressure) increases by ≥15% from the reference condition established during the first 48 h of operation. Cleaning procedures are usually given by the membrane manufacturers.17
In this case, the maximum operating pressure required is 15.5 bar and maximum pressure drop is 1 bar increased to 16.5 and 1.5bar, respectively, without an increase in the standard permeate (flow rate 27.3 m3/d and TDS 50 mg/l). Frequent shutdowns of the plant were observed due to membrane fouling (permeate flow rate is 18.7 m3/d and TDS is 580 mg/l).
In this case, both acidic and alkaline cleaners can be used. Acid cleaning to remove mineral scale was done at pH 2 or lower with 0.2% (W) hydrochloric. Citric acid can also be used in the same concentration. Alkaline cleaning to remove organic fouling was done at pH 12, generally done with 0.1% (W) sodium hydroxide 24,34. After resolving the fouling problem, membranes are cleaned with the first option given by the manufacturer every six months and the cartridge filters are replaced every three months1.
Conclusion
In our case study iron was removed in groundwater before passing through reverse osmosis membranes as pretreatment technique to avoid membrane fouling. Different pretreatment techniques are done to remove iron and save the membrane.
Many processes affecting the iron removal from the groundwater are applied in this case study. From the performance comparison between conventional and specific pre-treatment methods, we concluded that, every applied method has advantages and disadvantages in application. The most suitable pretreatment technique for iron removal (concentration less than 5 mg/l) is a granular activated carbon (GAC) filter which has higher adsorption capacity and leads to low
operating cost.
Acknowledgments
The authors express their appreciation to Prof. Dr. Naglaa Ali and Prof. Dr. Yasser Moustafa, Egyptian Petroleum Research Institute (EPRI), for their assistance and revision of this paper. Also, the authors wish to acknowledge the assistance and support of this study by Eng. Mohamed Amer, General Manager of Water Engineering Technology Co. (WETCO), which agent for each products of Jacobi Carbon, Clack and DOW.
References
- Arras W., Ghaffour N., Hamou A., Desalination 235: 170, (2009).
- Bodalo-Santoyo A., Gomez-Carrasco J.L., Gomez-Gomez E. and Montesinos A.M., ,Desalination ,160 : 151, (2004).
- Wolf P.H., Siverns S.and Monti S.,Desalination, 182: 293, (2005).
- Ellis D., Bouchard C. and Lantagne G.,Desalination . 130: 255, (2000).
- Sharma S.K., Kappelhof J., GroenendijkM.and Schippers J.C., J. Water Supply Res.Technol., 50: 187, (2001).
- EC-Official Journal of the EuropeanCommunities Council Directive 98/83/EC L.330: 32, (1998).
- Madaeni S.S., Mohammadi T. andMoghadam M.K., Desalination 134: 77,(2001).
- Lindau J.and Jonsson A.S., J. of MembraneScience 87: 71, (1994).
- Redondo J.A. and Lomax I., Desalination,110 : 167, (1997).
- Clack Corporation, Anthracite data sheet form no. 2354, replaces form 1785, Part no.A8029, 3 (2011).
- Clack Corporation, Filter sand and graveldata sheet form no. 2352, replaces form 1824,Part no. A8071, 3 (2011).
- Filmtec Reverse Osmosis Membranes,BW30-4040, Technical bulletin 609-00350-804
- Chua K.T., Hawlader M.N.A. and Malek A.,Desalination , 159: 225, (2003).
- Headquarters, Department of the Army,Washington, D.C., Water DesalinationTechnical Manual, (1986).
- Voutchkov N., Desalination , 261: 354,(2010).
- Voutchkov N.and Dietrich J., Pilot testingalternative pretreatment systems forseawater desalination in Carlsbad,California, Proceedings of World Congressin Desalination and Reuse, InternationalDesalination Association, SP05-095,Singapore, 11-16, (2005).
- Filmtec Reverse Osmosis Membranes Technical Manual,( 2004).
- Prihasto N., Liu Qi. and Hyun Kim S.,Desalination, 249: 308, (2009).
- Lessard C., Ellis D., J. S &odes and C.Bouchard, Environment, 32, (1999).
- Clack Corporation, manganese greensanddata sheet form no. 2349, replaces form 1564,Part no. A8041, 3, (2011).
- Benefield, L. D. and Judkins, J. F. ,Process Chemistry for Water and WastewaterTreatment. Prentice-Hall (1982).
- Mourato D. and Smith C., Proc., 6th Workshopon Drinking Water, AQTE, Montr6al, 705-716,( 1994).
- Cote P., Mourato D. , C.Gungerich, Russell J.and Houghton E., ISWA Conference,Membranes in Drinking and Industrial WaterProduction, Amsterdam ( 1998).
- Filmtec Reverse Osmosis MembranesTechnical bulletin 609-22010/CH172-086-E.
- Rohm and Haas Amberlite IR120Na, PDS0210 A.1997.
- Clack Corporation, Activated carbon data sheet form no. 2348, replaces form 1795 &1564, Part no. A8009-12, 3, (2011).
- Cheremisinoff N.P., Handbook of Water andWastewater Treatment Technologies,Butterworth-Heinemann, Boston, USA, 138:140, ( 2002).
- Ahmad bin Jusoh, Cheng W.H., Low W.M. ,Ali Nora’aini and Megat Mohd Noor M.J.,Desalination, 182: 347, (2005) .
- Tam Y.K., Studying on Removal of Dye UsingGranular Activated Carbon, ScienceEngineering Department, Kolej University ofScience and Teknology Malaysia ( 2003).
- Rockstraw D.A. and Dastgheib S.A., Carbon,40: 1843 (2002).
- Keneth E.N. and Chang H.T., Effect ofSurface Functional Groups on the Coefficientof Freundlich Isotherm, Department ofChemical and Environmental Engineering,Illinois Institute of Technology, 1999, http://www.chee.lit.edu/ chang/index.html,Accessed 18 April ( 2004).
- Ratnayaka Don D., Malcolm J. Brandt andMichael Johnson K., Water Supply (Sixth Edition), CHAPTER 10: Specialized and Advanced Water Treatment Processes, 365:423, (2009).
- WHO ,Guidelines for Drinking-Water Quality,Vol. 1, Recommendations. 2nd Edn (1993).
- Filmtec Reverse Osmosis MembranesTechnical bulletin 609-24010/CH172-120-E.







