A Hybrid Approach for Industrial Wastewater Remediation: Integrating Hydrodynamic Cavitation with Advanced Oxidation Processes.
1
Department of Chemical Engineering,
T.K.I.E.T.,
Warananagar,
Maharashtra
India
2
Nanasaheb Mahadik College of Engineering,
Peth Naka,
Sangali,
Maharashtra
India
3
Department of Technology,
Shivaji University,
Kolhapur,
Maharashtra
India
Corresponding author Email: arvindpatil2899@gmail.com
DOI: http://dx.doi.org/10.12944/CWE.20.2.26
Copy the following to cite this article:
Patil A. V, Anekar S. V, Patil P. P. A Hybrid Approach for Industrial Wastewater Remediation: Integrating Hydrodynamic Cavitation with Advanced Oxidation Processes. Curr World Environ 2025;20(2). DOI:http://dx.doi.org/10.12944/CWE.20.2.26
Copy the following to cite this URL:
Patil A. V, Anekar S. V, Patil P. P. A Hybrid Approach for Industrial Wastewater Remediation: Integrating Hydrodynamic Cavitation with Advanced Oxidation Processes. Curr World Environ 2025;20(2).
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2025-07-10 |
|---|---|
| Accepted: | 2025-09-01 |
| Reviewed by: | 
 Chadetrik Rout
Chadetrik Rout
|
| Second Review by: |

 Prashant Dhanake
Prashant Dhanake
|
| Final Approval by: | Dr. Gangadhar Andaluri |
Introduction
The dairy industry produces large volumes of wastewater characterized by high chemical oxygen demand (COD), biochemical oxygen demand (BOD5), total dissolved solids (TDS), fats, oils, greases, nitrogen, and phosphorus.1-4 These contaminants pose significant environmental challenges, including toxicity, eutrophication, and pollution of water bodies.5-6 The high variability in flow rates and pollutant concentrations across dairy operations and seasons further complicates treatment efforts.7-8 Treating this wastewater to meet stringent discharge regulations remains a critical and ongoing challenge for sustainable dairy processing.
Conventional biological treatments, including anaerobic reactors such as Upflow Anaerobic Sludge Blanket (UASB) and aerobic systems like sequencing batch reactors (SBR), are widely employed.3-6,9 Anaerobic treatment efficiently reduces organic load and generates biogas but often fails to fully degrade lipids and recalcitrant organics. Aerobic treatments can further reduce these pollutants but require high energy input and produce significant biomass.3-6,9 Advanced oxidation processes (AOPs) like ozonation and hydrogen peroxide treatment offer powerful oxidative degradation but are limited by cost and operational complexity.10-12 Thus, no single treatment method achieves complete removal efficiently and economically.13-14 The final effluent revealed that certain parameters, such as BOD5 and COD, had higher values than the intended levels as per the pollution control standards indicated for the dairy industry, even though the aforementioned techniques are utilized in the industry for treatment.15 Hence it needs to give more attention and treat the effluent until it gets the desired values. For the rehabilitation of contaminated ground, surface, and wastewaters containing non-biodegradable organic contaminants, advanced oxidation procedures (AOP) hold great promise.10-14 If the resultant intermediates are readily degradable in a subsequent biological treatment, then using AOP as a pretreatment step to improve the biodegradability of wastewater containing resistant or inhibiting chemicals can be justified.16 The influent needs to be pretreated and to improve its biodegradability to make it biodegradable for further biological processes.16 The biodegradability index (BI) is an indicator that quantifies the biodegradability of waste water based on the ratio of BOD5/COD. More the biodegradability index favors the amenability to biological treatment which leads to efficient biological treatment. Hence the Victor Sarria has suggested improving the BI by using processes like advanced oxidation processes (AOPs) evolves as one of the pretreatment options.16
Hydrodynamic cavitation (HC) generates intense localized energy and reactive hydroxyl radicals (.OH), which enhance pollutant breakdown.17 However, HC alone yields moderate treatment efficiencies due to limited cavitation intensity. Combining HC with AOPs (H2O2 or O3) can exploit synergistic effects, increasing radical generation and degradation rates.18-23 While prior studies explored binary combinations, the potential of ternary hybrids remains under-investigated, especially using real dairy wastewater.
Despite promising findings, existing literature mostly focuses on simulated or low-strength effluents and binary HC+AOP treatments. There is a clear gap in evaluating optimized ternary HC+H2O2+O3 systems on actual dairy effluent. This study aims to evaluate the impact of the ternary combination on improving the biodegradability index (BI), facilitating more efficient downstream biological treatment.
Materials and Methods
Materials
A nearby dairy company in Kolhapur, India, provided the actual industrial wastewater. The waste water received from all sections of the dairy was collected in the ETP from where the samples were collected. To account for variability in effluent composition, composite samples were collected over multiple days covering different shifts and production cycles. The 20 litre carboy was used to collect it. Wastewater is available in the milk and milk products processes along with washing and cleaning operations. Table 1 lists the wastewater sample's characteristics. The effluent mainly contained high levels of COD, BOD5, total dissolved solids (TDS) also oil and grease are characteristics of the dairy waste water. After then it was circulated through the hydrodynamic cavitation set up. The chemicals utilized in the project, including hydrogen peroxide, were acquired from S.D. Fine Chem. Pvt. Ltd. in Mumbai, India.
To determine BOD5 and COD all necessary AR grade chemicals were utilized in accordance with the APHA, 1998 standard protocol.24
Table 1: Characteristics of dairy wastewater.
Parameter | Observed Values | Units |
pH | 6.4 | - |
color | Milky white | - |
COD | 2745 | mg/L |
BOD5 | 964 | mg/L |
TDS | 680 | mg/L |
TSS | 90 | mg/L |
BOD5:COD ratio | 0.35 | - |
Oil & Grease (%) | 0.07 | % |
Experimental setup
An orifice plate acts as the main cavitating device in the Hydrodynamic Cavitation (HC) Reactor used in the present research, as shown in Figure 1. The reactor primarily operates in recirculation mode, where a 1 hp pump transfers liquid from a holding tank to both the main and bypass flow lines, with control valves regulating the flow rates. The orifice plate, which serves as the cavitation-inducing element, has a 3 mm diameter with three perforations. The geometric details of this orifice plate configuration have been documented in previous studies.25 A 15 L storage tank with a 20 L full capacity, equipped with a helical cooling coil for temperature regulation. The cooling coil is connected to an external tap water system for maintaining a constant wastewater temperature of 30°C. Flow control valves were installed on both the main and bypass lines to regulate the volumetric flow through the orifice, thereby controlling the cavitation intensity. Three control valves (V1–V3) were incorporated to regulate the pump's inlet and outlet flow. Pressure gauges were installed upstream (P1) and downstream (P2) of the orifice plate to measure pressure differential, which was used to calculate the cavitation number (Cv), an important dimensionless parameter indicating cavitation intensity. Pressure gauges (P1 and P2) were installed at the orifice plate's upstream and downstream directions to monitor the pressure differentials across the cavitating device. The orifice plate is affixed within the main flow line using flange arrangements. For the treatment involving ozone (O3), an ozone generator of 200 mg/hr capacity was employed.
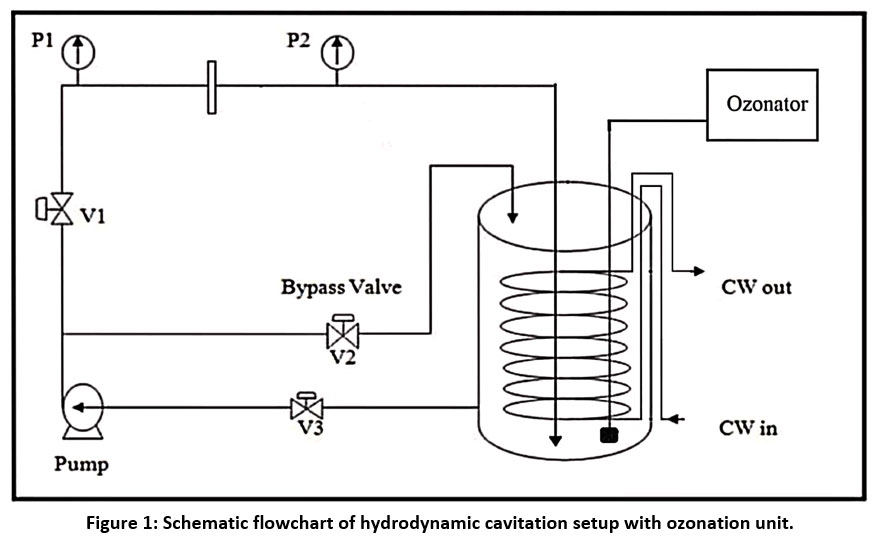 | Figure 1: Schematic flowchart of hydrodynamic cavitation setup with ozonation unit.
|
Procedure for experimentation and analysis-
Each experiment with the HC reactor was performed using 15 L of effluent for a total duration of 240 min (4 h). Samples were withdrawn at 60 min intervals to monitor changes in the biodegradability index. The impact of hydrodynamic cavitation (HC) in conjunction with hydrogen peroxide (H2O2) was assessed under ideal circumstances of 2 bar intake pressure. The influence of H2O2 loading was studied over a concentration range of 3–12 g/L. A ceramic diffuser was used to deliver ozone gas into the feed tank in order to promote the oxidative destruction of pollutants.
Treatments were conducted for the following conditions:
HC alone
HC combined with H2O2 and H2O2 alone at varying loadings (3, 6, 9, 12 g/L with HC and 9g/L without HC)
HC combined with ozone (O3) and ozone alone at a constant flow rate of 200 mg/h delivered via a ceramic diffuser into the feed tank
Ternary combination of HC + H2O2 (9 g/L) + O3 (200 mg/h)
Wastewater samples were collected from a dairy effluent treatment facility. The HC reactor setup was thoroughly washed with water and alkali to remove any residual contaminants. The wastewater was brought to room temperature (~30°C) from its storage temperature of 4–5°C before treatment. A cooling coil system installed inside the holding tank kept the wastewater temperature at 30°C. Standard operating procedures (SOPs), in accordance with traditional APHA methodologies, were used to examine Chemical Oxygen Demand (COD) and Biochemical Oxygen Demand (BOD5).24 After centrifuging the input samples, the BOD5 and COD were determined. Similar techniques of analysis were used for the treated wastewater. The Bio-Degradability index (BI) is computed using the obtained BOD and COD values. To monitor improvements in BI, samples were periodically withdrawn during the course of treatment.
Results
To attain objectives of the research work, experimental work was carried out for the hydrodynamic cavitation system alone and in combination with other chemical oxidants like H2O2 and ozone. All parameters were optimized to increase the biodegradability of the dairy waste water. The purpose of this study is to examine the impact of HC and other oxidants to improve the biodegradability index of dairy waste water before the biological processes in wastewater treatment. Each experiment was carried out with the HC reactor working at optimum circumstances, which included an inlet pressure of 2 bar and orifice plate having diameter of 3 mm for 3 no. of holes which was established in earlier work.25 All experimental treatments were carried out in triplicate to ensure reproducibility, and data are reported as mean values with standard deviations shown as error bars in Figures 2–4. The consistency in results across replicates confirms the reliability of the observed improvements in biodegradability index (BI). Statistical analysis using ANOVA confirmed that increases in BI due to combined treatments were significant (p < 0.05) compared to HC alone or oxidants alone.
Combined HC + H2O2 Treatment
This study highlights the impact of hydrogen peroxide (H2O2) on improving the biodegradability index (BI) of dairy wastewater, both with and without hydrodynamic cavitation (HC). The experiments were conducted using an orifice plate of 3 no. of holes with a diameter of 3 mm each at a temperature of 30°C and a pressure of 2 bar. H2O2 loadings of 3, 6, 9, and 12 g/L were tested, and the optimal BI improvement from 0.35 to 0.74 was achieved at 9 g/L as shown in fig. 2. Beyond this, at 12 g/L, the improvement was lower. Experiments with only H2O2 (bypassing the cavitation chamber) showed significantly lower BI improvement, reaching only 0.54 at 9 g/L. All experiments show the value stabilized after 180 minutes of treatment.
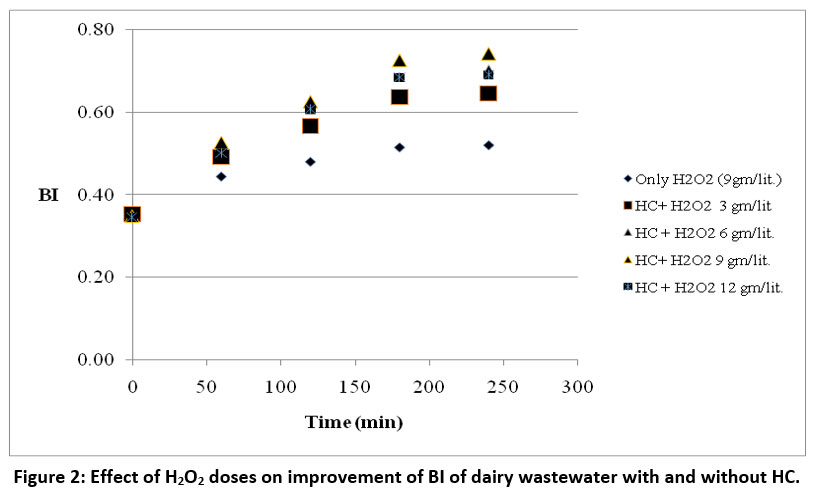 | Figure 2: Effect of H2O2 doses on improvement of BI of dairy wastewater with and without HC.
|
Combined HC + Ozone Treatment
By injecting 200 mg/hr of ozone straight into the feed tank, the impact of adding ozone to the HC was investigated. An orifice plate with three holes with a diameter of 3 mm each were used in the trials, which were carried out at 30°C and two bars of pressure. The results presented in Figure 3 demonstrate that the BI improvement achieved with the individual ozone strategy was amplified with the combined HC+O3 approach. The actual improvement in BI being from 0.35 to 0.58 and 0.35 to 0.81 respectively for only ozone and HC+O3 treatment. When compared to the only HC, the readings for the HC+O3 combination strategy were likewise higher.
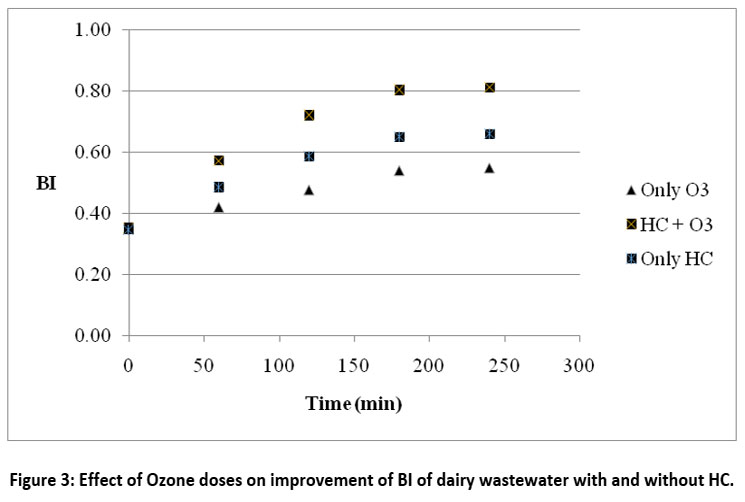 | Figure 3: Effect of Ozone doses on improvement of BI of dairy wastewater with and without HC.
|
Combined HC + H2O2 + Ozone (Ternary Combination)
It has been studied how the biodegradability index is affected by the combined effects of HC, H2O2 (optimal loading of 9 g/L), and O3 (flow rate 200 mg/hr). An orifice plate with three holes with a diameter of 3 mm each was used in the trials, which were carried out at 30°C and 2 bar of pressure. The biodegradability index has been improved from 0.35 to 0.89 using a combined strategy of HC, H2O2 (optimal loading of 9 g/L), and O3 (flow rate 200 mg/h), as shown in Fig. 4.
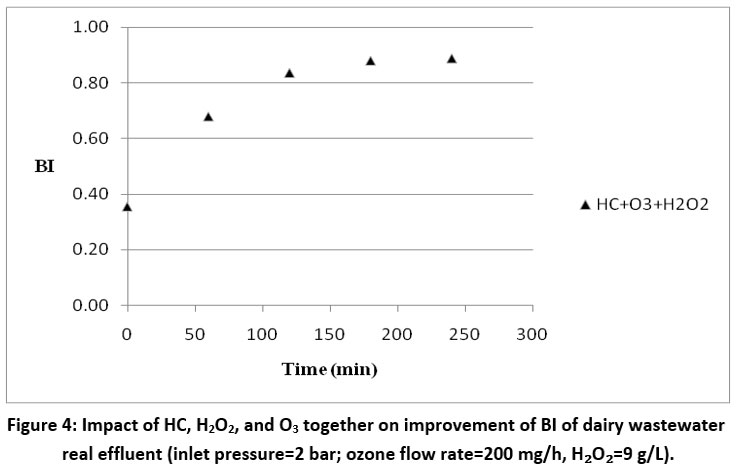 | Figure 4: Impact of HC, H2O2, and O3 together on improvement of BI of dairy wastewater real effluent (inlet pressure=2 bar; ozone flow rate=200 mg/h, H2O2=9 g/L).
|
Discussion
Combined HC + H2O2 Treatment
The dissociation of H2O2 due to cavitation, which increased the formation of hydroxyl radicals (•OH) and improved mass transfer, was credited with the better performance of HC coupled with H2O2. Supporting studies showed similar trends: Barik and Gogate reported optimal degradation of 2,4,6-trichlorophenol (TCP) at an H2O2 ratio of 1:5, while further increases led to diminishing returns.26 Dhanke’s research on vegetable oil residue wastewater (VORWW) found an optimal H2O2 concentration of 25 g/L, beyond which improvements in BI were minimal.27 Likewise, in pulp and paper effluent, raising H2O2 dosages from 111 to 222 mg/L improved COD reduction; but, at higher concentrations, hydroxyl radical depletion resulted in diminishing results. These findings underscore the importance of optimizing H2O2 dosing for effective wastewater treatment using HC.28
Combined HC + Ozone Treatment
The depletion of ozone molecules due to cavitational effects, which eventually increases the availability of OH radicals, is the cause of the greater degree of degradation for the HC+O3 combination. The synergistic interaction between cavitation-induced turbulence and the oxidative potential of ozone is responsible for the increased degradation efficiency shown in the combined hydrodynamic cavitation (HC) and ozone (O3) treatment procedure.
Cavitation increases the production of reactive species such hydroxyl radicals (•OH) and nascent oxygen by facilitating the breakdown of ozone molecules, which significantly contribute to pollutant breakdown in aqueous systems.26 Raut-Jadhav et al. showed that when applied to pesticide industry effluent, HC and ozone at a dosage of 3 g/h produced a roughly fivefold higher COD reduction than HC alone.29 Similarly, Gogate and Patil reported that for triazophos degradation, the combined HC+O3 process achieved up to 86.68% degradation when ozone was directly introduced into the feed tank, outperforming the 78.12% degradation observed when ozone was injected through the orifice. In contrast, individual treatments with only HC and only ozonation yielded 49.67% and 59.36% degradation, respectively.30 These findings collectively affirm the superior efficiency of HC–ozone hybrid systems in degrading persistent organic pollutants.
Combined HC + H2O2 + O3 (Ternary Combination)
Thanekar et al. studied how hydrogen peroxide (H2O2), ozone (O3), and hydrodynamic cavitation (HC) worked together to lower the chemical oxygen demand (COD) in industrial effluent. The study reported a COD reduction of 60.8% when all three advanced oxidation processes were applied simultaneously, which was significantly higher than the reduction achieved by each process when used individually. The enhanced performance was attributed to the synergistic interaction among HC, H2O2, and O3, leading to the formation of a greater concentration of hydroxyl radicals (•OH). These highly reactive species effectively degrade complex organic compounds present in the wastewater, resulting in improved treatment efficiency.31
Conclusion
The current study shows how hydrodynamic cavitation (HC) can be used to treat industrial wastewater both alone and in conjunction with advanced oxidation processes (AOPs) such as ozone (O3) and hydrogen peroxide (H2O2). A comprehensive review of literature provided key insights into the selection and optimization of HC reactor operating parameters and synergistic effects with oxidants. Experimental investigations using real industrial effluent revealed that HC alone enhanced the biodegradability index (BI) from 0.35 to 0.66. This improvement increased to 0.74 with the addition of H2O2 (9 g/L), and further to 0.81 when ozone (200 mg/h) was used in combination with HC. The highest enhancement was achieved using a ternary combination of HC + H2O2 + O2, resulting in a BI increase from 0.35 to 0.89, indicating significant synergistic interaction.
The generation of reactive hydroxyl radicals (•OH) through cavitation and oxidation reactions plays a critical role in breaking down complex organic pollutants into simpler, less toxic compounds. This increases the biochemical oxygen demand (BOD5) and decreases the chemical oxygen demand (COD), making the effluent more biodegradable. The results clearly establish that the combined HC + H2O2 + O2 treatment is more effective than the individual or binary combinations and offers a promising pre-treatment strategy for improving the efficiency of downstream biological processes. Therefore, this approach can significantly contribute to the sustainable management of dairy and industrial wastewater.
Acknowledgement
I would like to thank Prof. S.V. Anekar for the useful suggestions and guidance provided during this research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) declares no conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources:
Not Applicable
Author Contributions-
Arvind Vilas Patil- Conceptualization, Methodology, Data Collection, Analysis, Project Administration, Writing – Original Draft, Writing – Review & Editing,
Sunil Vamanrao Anekar- Supervision
Prashant Pandurang Patil- Supervision
References
- Sharma H, Singh PK, Kaur I, Singh R. Water footprints of dairy milk processing industry: A case study of Punjab (India). Water. 2024;16(3):435. doi:10.3390/w16030435.
CrossRef - Al-Tayawi AN, Sisay EJ, Beszédes S, Kertész S. Wastewater treatment in the dairy industry from classical treatment to promising technologies: An overview. Processes. 2023;11(7):2133. doi:10.3390/pr11072133.
CrossRef - Al-Qodah Z, Al-Shannag M, Hudaib B, Bani-Salameh W. Enhancement of dairy wastewater treatment efficiency in batch chemical-assisted solar-powered electrocoagulation-adsorption system. Case Stud Chem Environ Eng. 2024;9:100760. doi:10.1016/j.cscee.2024.100760.
CrossRef - Velmurugan L, Pandian KD. Enhancing physico-chemical water quality in recycled dairy effluent through microbial consortium treatment. Heliyon. 2024;10(21):e39501. doi:10.1016/j.heliyon.2024.e39501.
CrossRef - Kaur N. Different treatment techniques of dairy wastewater. Groundw Sustain Dev. 2021;14:100640. doi:10.1016/j.gsd.2021.100640.
CrossRef - Ahmad T, Aadil RM, Ahmed H, et al. Treatment and utilization of dairy industrial waste: A review. Trends Food Sci Technol. 2019;88:1-14. doi:10.1016/j.tifs.2019.03.030.
CrossRef - Patil NS, Kurhekar JV. An investigation of monthly variation of different physicochemical parameters of dairy industry effluent. Sustain Agri Food Environ Res. 2022;10(1):20-26. doi:10.7770/safer-V10N1-art2519.
CrossRef - Leena AV, Meiaraj C, Balasundaram N. BOD/COD a measure of dairy waste treatment efficiency – a case study. IOSR J Mech Civ Eng. 2016;13(5, ver VIII):107-114.
- Joshiba GJ, Kumar PS, Femina CC, et al. Critical review on biological treatment strategies of dairy wastewater. Desalin Water Treat. 2019;160:94-109. doi:10.5004/dwt.2019.24194.
CrossRef - Hubner U, et al. Advanced oxidation processes for water and wastewater treatment – guidance for systematic future research. Heliyon. 2024;10(9):e30402. doi:10.1016/j.heliyon.2024.e30402.
CrossRef - Thanekar P, Panda M, Gogate PR. Degradation of carbamazepine using hydrodynamic cavitation combined with advanced oxidation processes. Ultrason Sonochem. 2018;40(pt A):567-576. doi:10.1016/j.ultsonch.2017.08.001.
CrossRef - Wang Z, Manickam S, Wang W, Fu L, Bie H, Wang B, Sun X. Hybrid advanced oxidation process for rapid ciprofloxacin removal: Coupling hydrodynamic cavitation with UV/H2O2. Ultrason Sonochem. 2025. doi:10.1016/j.ultsonch.2025.107475.
CrossRef - Bai X, Li C, He S, Zhou J, Hu J. Combining advanced oxidation processes with biological processes in organic wastewater treatment: Recent developments, trends, and advances. Desalination and Water Treatment. 2025;323:101263. doi:10.1016/j.dwt.2025.101263.
CrossRef - Yeneneh AM, Al Balushi K, Jafary T, Al Marshudi AS. Hydrodynamic cavitation and advanced oxidation for enhanced degradation of persistent organic pollutants: A review. Sustainability. 2024;16(11):4601. doi:10.3390/su16114601.
CrossRef - Central Pollution Control Board (CPCB), Government of India. The Environment (Protection) Rules, 1986, Schedule VI: General Standards for Discharge of Environmental Pollutants. 1993, amended 2009.
- Sarria V, Parra S, Adler N, Péringer P, Benítez N, Pulgarín C. Recent developments in the coupling of photoassisted and aerobic biological processes for the treatment of biorecalcitrant compounds. Catal Today. 2002;76(2-4):301-315. doi:10.1016/S0920-5861(02)00206-5.
CrossRef - Raut-Jadhav S, Saini D, Sonawane S, Pandit A. Effect of process intensifying parameters on the hydrodynamic cavitation-based degradation of commercial pesticide (methomyl) in the aqueous solution. Ultrason Sonochem. 2016;28:283-293. doi:10.1016/j.ultsonch.2015.08.004.
CrossRef - Merchant AI, Vakili AH, Kocaman A, Abu Amr SS. New advancement of advanced oxidation processes for the treatment of petroleum wastewater. Desalin Water Treat. 2024;319:100565. doi:10.1016/j.dwt.2024.100565.
CrossRef - Ma D, et al. Critical review of advanced oxidation processes in organic wastewater treatment. Chemosphere. 2021;275:130104. doi:10.1016/j.chemosphere.2021.130104.
CrossRef - Rekhate CV, Srivastava JK. Recent advances in ozone-based advanced oxidation processes for treatment of wastewater – a review. Chem Eng J Adv. 2020;3:100031. doi:10.1016/j.ceja.2020.100031.
CrossRef - Asheghmoalla M, Mehrvar M. Photodegradation of aqueous pharmaceuticals in a continuous UV/H2O2 system: Photoreactor modeling. J Environ Chem Eng. 2025;13:117115. doi:10.1016/j.jece.2025.117115.
CrossRef - Raut-Jadhav S, Saharan VK, Pinjari DV, Saini DR, Sonawane SH, Pandit AB. Intensification of degradation of imidacloprid in aqueous solutions by combination of hydrodynamic cavitation with various advanced oxidation processes (AOPs). J Environ Chem Eng. 2013;1(4):850-857. doi:10.1016/j.jece.2013.07.029.
CrossRef - El-Magied El-Sonbati MA, El-Batrawy O, El-Awadly EM, Hegazy T. Pretreatment of high organic load dairy industry wastewater by chemical coagulation and advanced oxidation processes. Catrina Int J Environ Sci. 2020;20(1):53-60. doi:10.21608/cat.2020.94172.
CrossRef - APHA (American Public Health Association). Standard Methods for the Examination of Water and Wastewater. 20th ed. APHA; 1998.
- Patil AV, Anekar SV, Patil PP. Unlocking biodegradability: pretreatment of dairy industry wastewater by hydrodynamic cavitation. Curr World Environ. 2025;20(1):299-309. doi:10.12944/CWE.20.1.23.
CrossRef - Barik AJ, Gogate PR. Degradation of 4-chloro 2-aminophenol using combined strategies based on ultrasound, photolysis and ozone. Ultrason Sonochem. 2016;28:90-99.
CrossRef - Dhanke P, Wagh S. Treatment of vegetable oil refinery wastewater with biodegradability index improvement. Mater Today Proc. 2020;27(1):181-187.
CrossRef - Asha A, Keerthi, Muthukrishnaraj A. Improvement of biodegradability index through electrocoagulation and advanced oxidation process. Int J Ind Chem. 2014;5:4.
CrossRef - Raut-Jadhav S, Badve MP, Pinjari DV, Saini DR, Sonawane SH, Pandit AB. Treatment of the pesticide industry effluent using hydrodynamic cavitation and its combination with process intensifying additives (H2O2 and ozone). Chem Eng J. 2016;295:326-335.
CrossRef - Gogate PR, Patil PN. Combined treatment technology based on synergism between hydrodynamic cavitation and advanced oxidation processes. Ultrason Sonochem. 2015;25:60-69.
CrossRef - Thanekar P, Gogate PR. Combined hydrodynamic cavitation-based processes as an efficient treatment option for real industrial effluent. Ultrason Sonochem. 2019;53:202-213.
CrossRef
Abbreviations List
HC - Hydrodynamic Cavitation,
AOPs - Advanced Oxidation Processes,
BI - Biodegradability Index,
COD - Chemical Oxygen Demand,
BOD5 - Biochemical Oxygen Demand (5-day),
TDS - Total Dissolved Solids,
TSS - Total Suspended Solids,
ETP - Effluent Treatment Plant,
APHA - American Public Health Association.
TCP - 2,4,6-Trichlorophenol,
VORWW - Vegetable Oil Residue Wastewater,
SBR - Sequencing Batch Reactor,
UASB - Upflow Anaerobic Sludge Blanket,
SOPs - Standard Operating Procedures,
ANOVA- Analysis of Variance






