Contamination of Heavy Metals and Physicochemical Parameters of Water and Sediment in Haramaya Lake, Ethiopia
Yaecob Gebre Leleko12
 , Sissay Menkir2
, Sissay Menkir2
 , Endale Teju3
, Endale Teju3
 , Ranjeeta Bhari1
, Ranjeeta Bhari1
 and Balwinder Singh Sooch1
*
and Balwinder Singh Sooch1
*

1
Department of Biotechnology and Food Technology,
Punjabi University,
Patiala,
Panjab
India
2
Departments of Biology and Biotechnology,
Haramaya University,
Dire Dawa,
Ethiopia
3
Department of Chemistry,
Haramaya University,
Dire Dawa,
Ethiopia
Corresponding author Email: soochb@pbi.ac.in
DOI: http://dx.doi.org/10.12944/CWE.20.2.19
Copy the following to cite this article:
Leleko Y. G, Menkir S, Teju E, Bhari R, Sooch B. S. Contamination of Heavy Metals and Physicochemical Parameters of Water and Sediment in Haramaya Lake, Ethiopia. Curr World Environ 2025;20(2). DOI:http://dx.doi.org/10.12944/CWE.20.2.19
Copy the following to cite this URL:
Leleko Y. G, Menkir S, Teju E, Bhari R, Sooch B. S. Contamination of Heavy Metals and Physicochemical Parameters of Water and Sediment in Haramaya Lake, Ethiopia. Curr World Environ 2025;20(2).
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2025-05-20 |
|---|---|
| Accepted: | 2025-08-08 |
| Reviewed by: | 
 Kavach Mishra
Kavach Mishra
|
| Second Review by: |

 Shah, Ahsan
Shah, Ahsan
|
| Final Approval by: | Dr. Gopal Krishan |
Introduction
Water is one of the most essential natural resources.1,2 Despite the fact that water covers roughly 71% of the Earth’s surface, seawater make up 97% of it and is generally unsuitable for human use.3 Only 3% of the world's population depends on fresh water, as seen by the worldwide water distribution, because seawater is rarely suitable for human use.4 The only 0.06% of the freshwater that is available is readily accessible; the remainder is made up of groundwater, rivers, and the frozen polar ice cap or glaciers. In the global ecology, lakes and rivers are important because they provide drinking water, industrial water, shipping water, fish farming water, and irrigation water.3 Currently, with the increasing demand for water for domestic, industrial, and agricultural purposes brought on by population growth, water is becoming a scarce resource globally.5 It is projected that between 1.4 and 2.1 billion people worldwide are under insufficient water resources. This state is explained by either a per capita water availability of less than 1000 m3 or a long-term average annual runoff of greater than 0.4.6 The freshwater ecosystems are becoming more and more degraded. Human activities add to the natural stresses that organisms encounter in their surroundings.7 Thus, improving our knowledge of freshwater ecology is essential for both its biological relevance and efficient freshwater resource management, both of which are critical to human welfare. Clean water is essential for minimizing disease and enhancing a good quality of life. All living things on Earth need water to survive, both; water life and humans depend on it.8
However, the sanitation of lake and river water has polluted as a result of human population development and careless water resource use.9 The health management of water resources are further jeopardized by the increased pollution burden caused by toxic wastewater and runoff from municipal, industrial & agricultural sources.10 Thus, knowledge of surface water quality is essential for several uses, such as agriculture, industry, and drinking.11 Aquatic creatures are seriously threatened by pollution in aquatic habitats.12 This contamination comes from a variety of sources, especially farming operations that release pesticides into freshwater bodies.13 The transportation of sewage, fertilizers, and pesticides by agricultural runoff contributes considerably to the introduction of heavy metals in to the aquatic environments and sediments.14 Numerous aquatic organisms may be impacted by heavy metal pollution, which can also negatively impact ecological equilibrium.15 Due to their inability to biodegrade and their ability to accumulate in the tissues of a wide range of organisms, heavy metal contamination has become a significant issue in aquatic environments.16,17 The primary health issues are the elevated levels of lead, cadmium, copper, mercury, arsenic, and selenium in water bodies, which are vital for the agriculture of the local population.18 Cadmium primarily affects the kidney, contributing to chronic kidney disease and reduced renal function.19 Similar to Parkinson’s disease, manganese overexposure can damage the brain, leading to neurological disorders, particularly in occupational settings. While in adults, lead contributes to kidney damage and hypertension, harmful to children, impairing cognitive development and lowering IQ. The copper element, though essential in small amounts, can cause gastrointestinal distress and, in chronic cases, liver and kidney toxicity, especially in genetic disorders. Throughout Southeast Asia, particularly in Bangladesh, India, and the Bengal region, eating rice and drinking water contaminated with heavy metals like arsenic has been connected to a range of health issues. These issues include cancers of the skin, kidney, bladder, and lungs.19 As seen in China, eating foods tainted with selenium has been connected to health problems such as osteoporosis, brittle nails, and hair loss.20 Moreover, elevated nickel and copper levels in food and water have been connected to liver and renal failures.21 The effective organization of water possessions requires an understanding of the individual contaminants present in a region, as well as the point source of contamination.
Metal pollution is increasing in several Ethiopian Rift Valley Lakes, primarily as a consequence of increased industrial activity and more intensive farming methods.22 Heavy metals are among the pollutants that can build up and be biomagnified through the food chain under specific environmental conditions, possibly reaching dangerous levels and harming the environment.23 Therefore, these heavy metals were selected for this study because of their toxicity, health hazards, and environmental persistence. While cadmium and lead are non-essential and extremely poisonous, copper and manganese are essential trace elements that become hazardous in high concentrations.24 These metals commonly originated from industrial, agricultural, and urban sources, and they can accumulate in water and sediments, posing serious pressure to water environment and human health condition. Their inclusion helps to assess pollution levels and ecological risk.25,26 This study offers a comprehensive assessment of pollution in Haramaya Lake by simultaneously analyzing heavy metals and physicochemical parameters in both water and sediment. Unlike previous research that focused on a single medium, this approach provides a holistic understanding of contamination. The findings fill a critical research gap, establish baseline data, and highlight potential ecological and health risks, contributing valuable insights for environmental monitoring and sustainable management.
Materials and Methods
Study area
The Haramaya area of the east Hararghe Zone in the Oromia region in eastern Ethiopia is home to Lake Haramaya. The lake lies approximately 505 kilometers east of Addis Ababa, 14 kilometers northwest of Harar, and 38 kilometers west of Dire Dawa. Geographically, it lies between latitudes 9°23'18" & 9°26'48" North and longitudes 41°58'30" and 42°05'30" East. Climatically, the region is characterized by a two modes of rainfall distribution, receiving an average yearly precipitation of approximately 751 mm. The Woreda boasts 2,424 improved groundwater wells, 24,827 traditional wells, and 2,948 ponds. These water sources support approximately 21,400 farm households in achieving food self-sufficiency and improving their livelihoods.21 The practices underscore the community's reliance on both crop cultivation and livestock rearing, supported by strategic irrigation efforts to optimize agricultural output. The Lake Haramaya watershed is a highly cultivated region with minimal arable land left for fallow and private grazing.27 The area's topography is predominantly undulating and rolling, covering approximately 71% of the watershed, while hilly and steep lands in the east and northeast account for only 8%. The remaining 17% consists of flat land situated in the middle and near the catchment. The altitude within the watershed varies significantly, ranging from 1,480 to 2,343 meters. Due to cultivation on sloping lands, soil erosion has become a critical environmental issue.28
The slope characteristics indicate that undulating and rolling terrain dominates, covering 74% of the catchment, with hilly and steep areas making up 8%, and flat land contributing to 17%. Additionally, convex and concave landforms are nearly equal, each comprising 47% of the total area, while land with a uniform slope represents only 6%. Administratively, Lake Haramaya is situated at the junction of five kebeles: Amuma, Kura Jalala, Finkile, Tinike, and Tuji Gabisa farmers' associations.28 The lake itself covers approximately 47 hectares, while 1,790 hectares have been identified as priority development areas for irrigation. Of this, around 880 hectares are currently irrigated using traditional flood and furrow methods, while an additional 580 hectares benefit from shallow groundwater and well irrigation schemes. Despite the total of 4,636 households with access to irrigation, only 2,720 households are directly benefiting from the 1,460 hectares of irrigable land. This highlights the need for improved irrigation infrastructure and equitable access to water resources to support the region's agricultural productivity and sustainability.29
Equipment and Chemicals
Ceramic pestle and mortar (catalog no. 7722-500, 500 ml) were used to grind and homogenize dried sediment samples. Solid or powder chemicals were weighed using a digital analytical balance (model BLS ew2001) and dried in a dry heat oven. A flame atomic absorption spectrophotometer (FAAS) (AAnalyst 400 model) was used to analyze the samples after they had been digested, cooled, filtered, and diluted with double-distilled water.30 Borosilicate volumetric flasks were used for dilutions and standard preparations. Sample digestion was performed using hot plates under a hood, and digested samples were stored in a refrigerator until further use. Metal standards were prepared from nitrate salts of copper, cadmium, lead, and manganese, and digestion employed 68% HNO3, 30% H2O2, and 37% HCl. All materials and reagents were used from Haramaya general laboratory and chemistry laboratory in the chemistry department of Haramaya University, Ethiopia.
Sample collection and analysis
The selection of sampling sites in a lake is guided by several key criteria to ensure comprehensive and representative data collection. In this study, three sampling sites were strategically chosen based on geographical distribution, anthropogenic influence, and ecological significance. Site one was selected near agricultural runoff zones, where the likelihood of nutrient and pesticide contamination is high due to farming activities. The second site was selected based on an urban area influenced by domestic wastewater discharge, representing potential pollution hotspots. A third site was located in the open water, away from the shore, to reflect the general and mixed conditions of the lake.
The necessary protocols were followed to ensure accuracy and prevent contamination during the collection of water and sediment samples for heavy metal analysis. Proper collection, preservation, and storage are crucial to obtaining reliable data for heavy metal contamination assessment.31 The calibration curve was created to determine the concentrations of copper, lead cadmium, and manganese in the experimental samples. To ascertain the levels of selected heavy metals in the experimental samples, a calibration curve was created. To produce four working standards for each metal ion of interest, a series of standard solutions were created using the stock solutions and diluted with distilled water.
Table 1: Comparing freshwater and sediment heavy metal levels to international standards.32-35
Heavy metals | Sample types | Concentration in uncontaminated water | WHO limit (mg/L or mg/kg) | FAO limit (mgL-1 or mgkg-1) | USEPA limit (mgL-1 or mg/kg-1) | Significant and risk |
Lead (Pb) | Water | 0.01 | 0.01 | 0.05 | 0.015 | Exceeds WHO and USEPA neurotoxic, affects kidney and blood stream |
Sediment | 35.0 mg/kg | 50 mg/kg (soil) | 35 mg/kg (threshold effect) | Exceeds limit: harmful to benthic organisms | ||
Cadmium (Cd) | Water | 0.03 | 0.03 | 0.01 | 0.005 | Slightly exceeds WHO: causes kidney/liver damage |
Sediment | 0.6 | 1 mg/kg | 0.99 mg/kg | Exceeds all: high bioaccumulation risk | ||
Copper (Cu) | Water | 1.9 | 2.0 | 0.02-0.5 | 1.3 | Exceeds all: gastrointestinal and liver effects |
Sediment | 35.7 | 35 mg/kg | 31.6 mg/kg | High levels: toxic to aquatic life | ||
Manganese (Mn) | Water | 0.1 | 0.1 | 0.2 | 0.05 (aesthetic) | Above limits: neurotoxic effects (children) |
Sediment | 1.1 | - | - | Elevated: possible oxidative stress in fauna |
Using a (FAAS), four metals in all were examined. The instrument's absorption mode was used to analyze all four metals (Cu, Cd, Pb, and Mn). The measured absorbance data and concentrations were used to create the calibration curve for each set of standard metal ion solutions. The unknown concentration of the samples was determined by using the formula from the standard curve.36
Physico-chemical analysis
The pH is measured using a pH meter, such as the HI5221 Benchtop pH Meter manufactured by Hanna Instruments, which accurately detects the hydrogen ion concentration in a solution and displays the pH value digitally, a thermometer is used to monitor the lake water’s temperature. A pH meter is calibrated using standard buffer solutions, typically starting with pH 7.00, then measuring each sample one by one. To avoid contamination of the samples, distilled water was used to rinse the electrode in between each buffer. Once the meter reading stabilizes in each buffer, it is adjusted or confirmed to match the known pH value. For greater accuracy, a third buffer can be used. Calibrations were done regularly to ensure accurate and reliable pH measurements. Electrical conductivity was determined with a JENWAY 4310 conductivity meter equipped with an electrode/ATC probe by applying a voltage between the electrodes submerged in 50 mL of the sample; the conductivity in µS/cm was then calculated based on the voltage drop due to water resistance.
Table 2: The world standard limit of the parameters in drinking water.37-39
Parameter | Parameters measured in uncontaminated water | Permissible Limit |
Temperature | 26 °C | 25-30 °C (desirable); not specified maximum |
pH | 6.9 | 6.5-8.5 |
Electrical conductivity (EC) | 0.76 mS/cm or 760 µS/cm | 500-1500 µS/cm (no fixed WHO limit, varies by region) |
Total dissolved solids (TDS) | 700 mg/L | Desirable: 500 mg/L; Maximum: 2000 mg/L |
The filtered water samples were evaporated for two hours at 105 °C in a 50 ml beaker that had been previously weighed in order to determine the total dissolved solids (TDS). After cooling the beaker for one hour, its final weight was recorded to calculate the TDS. Turbidity was first removed using Whatman filter paper, qualitative 150 mm grade-1, to ensure accurate TDS measurements.40
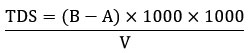
Where
TDS = Total dissolved solids (mg/L)
A = Initial weight of the evaporating beaker (g)
B = Final weight of the evaporating beaker (g)
V = volume of the sample (ml)
Indicators of Pollution Assessment
The levels of heavy metal in the samples were evaluated using a variety of quantitative criteria. Among these indices are:
Contamination Factor (CF)
Using the contamination factor, the level of heavy metal contamination in the sample was evaluated. The expression of CF:
![]()
Where, CF= contamination factor
Cn = amount of heavy metals in samples
Bn = environmental value of heavy metals in their natural state
Depends on the contamination factor, the heavy metals are classified as 41
CF < 1 is low
1 < CF < 3 is moderate
3 < CF < 6 is considerably high
CF > 6 is very high
Geo-accumulation index Igeo) analysis
![]()
Where HMc = reference the amounts of heavy metal, and HMs = heavy metals concentrations in the sample.
1.5 = constant Igeo = < 0: no pollution
Igeo = 0-1: moderate pollution Igeo = 1-2: strong pollution
Igeo = 2-3: high pollution Igeo = 3-4: very high pollution
Igeo = 4-5: severe pollution
Igeo = < 5: extreme pollution are the classifications utilized for the heavy metals.42,43
Pollution load index (PLI) assessment
The degree of contamination in water and sediment samples taken from various sampling locations of the lake was evaluated using the pollution load index. The expression for PLI is:
![]()
Where, PLI is the index of pollution load
CF is the contamination factor
n is the figure of elements
According to the report of the risk assessment for human health and environmental contamination states that, pollution is indicated by a PLI > 1, whilst no pollution is indicated by a PLI < 1.44
Ecological risk index (ERI)
The ecological or environmental risk index (ERI) for each metal is obtained by multiplying its toxicological response factor (Tr) with the contamination factor (CFn).
![]()
ERI’s classifications for heavy metals were:45
ERI>240 is significant environmental elevated risk
ERI 120-240 is high ecological risk
ERI 60-120 is considerably ecological risk
ERI 30-60 is reasonable ecological risk
ERI<30 is low ecological risk
Statistical data analysis
Leven’s test for equal variances (p<0.05) and the statistical package for social sciences (SPSS) software version 20 were used to assess the data for normality and homogeneity of variance. To compare concentration levels across the sites and sampling seasons (a = 0.05), ANOVA or analysis of variance was performed on the selected research sites using data that satisfied the normal assumptions. For data that did not satisfy the homogeneity of variance and normality criteria, non-parametric tests were employed.46
Results
Physico-chemical parameters of samples
Table 3 provides the mean values and standard deviations (±) for four essential physico-chemical analysis of the temperature (Temp), pH, electrical conductivity (EC), and total dissolved solids (TDS) measured at three different sampling sites during both the dry and wet seasons, with values compared against WHO guide line boundaries for safe fresh water.37,38 Water samples during the dry season from sites one, two, and three had mean temperatures of 24±0.003 oC, 24±0.006 oC, and 24±0.004 oC, respectively. Likewise, during the rainy season, the average temperature of water samples collected from sites one, two, and three was 27±0.003 oC, 27±0.003 oC, and 27±0.005 oC, respectively. During the dry season, all sampling sites recorded a uniform temperature of 24 oC, with extremely low variability, ranging from ±0.003 to ±0.006, indicating that the thermal regime across the study area was stable and unaffected by site-specific environmental variations.
Water samples taken during dry seasons from sites one, two, and three had mean pH values of 7.14±0.004, 7.12±0.005, and 7.15±0.003 (Table 3). The same sites had mean pH values of 7.13±0.003, 7.11±0.003, and 7.13±0.003 throughout the rainy season, respectively. It was found that the water samples from both the wet and dry seasons had rather than alkaline pH values. Table 3 displays a small (p<0.05) shift in the pH values of the water samples taken throughout both seasons.
Table 3: The standard deviation of the means ± for the physicochemical properties of water samples taken throughout both seasons.
Seasons | Sites | Temp | pH | EC | TDS |
Dry | site one | 24±0.003 | 7.14±0.004 | 1.55±0.003 | 569±0.003 |
site two | 24±0.006 | 7.12±0.005 | 1.3±0.006 | 541±0.006 | |
site three | 24±0.004 | 7.15±0.003 | 1.44±0.005 | 559±0.003 | |
MVDS | 24±0.004 | 7.14±0.004 | 1.43±0.005 | 556±0.004 | |
Wet | site one | 27±0.003 | 7.13±0.003 | 1.4±0.003 | 570±0.006 |
site two | 27±0.003 | 7.11±0.003 | 1.28±0.003 | 543±0.002 | |
site three | 27±0.005 | 7.13±0.003 | 1.36±0.003 | 562±0.003 | |
MVWS | 25±0.004 | 7.13±0.003 | 1.35±0.003 | 558±0.004 | |
OMVDWS | 25.5±0.004 | 7.13±0.003 | 1.4±0.004 | 557±0.004 | |
Maximum Permissible Limit | 25-30 °C *** | 6.5–8.5*** | 500-1500 µS/cm *** | 1000*** | |
During the dry season, the range of pH values was between 7.12 and 7.15, while during the wet season, they were between 7.11 and 7.13 (Table 3), suggesting that the water remained near neutral and chemically balanced. One of the most significant indicators of water quality and contamination levels in the selected areas under study is the pH value of aquatic systems, and the most crucial markers of pollution levels and the quality of water in the regions being studied. Uncontaminated water usually has a pH that is either slightly alkaline or almost neutral.47 Thus, when used for different purposes in the area, the lake water in this study does not provide any health risks to aquatic creatures or humans (Table 3). However, every pH value stayed within the range specified by the WHO recommendations, optimal for most aquatic organisms, irrigation, and watering cattle.
The water’s capacity to conduct electricity as results of dissolved ions is measured by its electrical conductivity (EC), which varied between 1.3 and 1.55 mS/cm at site two and one, respectively. In each season, the three sampling locations had different mean EC values. Similarly, the findings showed that the electrical conductivity (EC) values for the majority of the lake's sampling locations varied significantly (p < 0.05).
All results were failing within the acceptable range as per WHO, except site one, which was slightly exceeded. This can result from natural processes such as the dissolution of minerals in rocks and soil, human activities like agricultural runoff, industrial discharge, and wastewater input. Total dissolved solids (TDS) values, presenting overall concentration of organic and inorganic substances in the range of water from 541 to 569 mgL-1, well below the standard line of 1000 mgL-1. These results indicate moderate mineral content, safe for human consumption. The mean values for the dry season were recorded at 24±0.004 °C, pH 7.14±0.004, EC 1.43±0.005 mS/cm, and TDS 556±0.004 mg/L, further confirming the general consistency of water quality during the dry period. A comparable study found that TDS levels in the Huluka River in Ambo ranged from 351 mgL-1 during the dried out period to 551 mgL-1 during the wet period. On the other hand, TDS levels were higher than the WHO norm.34 This was probably brought about by agricultural operations like riverbank cutting, riverbed cultivation, fertilizer and pesticide use, and riverbank erosion.
Environmental Risk Evaluation
The different contamination assessment indices that were developed utilizing the heavy metal content of the sample and the estimated CF and ER for adults and children are shown in Table 4. Cadmium, manganese, copper, and lead had respective CF values of 13.8, 3.10, 0.78, and 1.12. Manganese and cadmium showed very high contamination factors, but copper and lead showed low contamination factors (CF < 1) and moderate contamination factors (CF < 3).
In heavy metal samples' pollution factor increased in the following sequence during both wet and dry seasons: Cu < Pb < Mn < Cd. While Cu indicates that the metals are not a source of pollution to the samples, PLI values for Cd, Mn, and Pb are greater than 1, indicating a sample that is contaminated with heavy metals. Igeo is used to assess the amount of metals in contaminated samples.48 In this investigation, the Igeo values for Cd, Mn, Cu, and Pb were 0.94, 0.37, -0.26, and -0.11 for the wet season, respectively. For dry seasons, the Igeo values for Cd, Mn, Pb, and Cu were 0.98, 0.37, -0.11, and -0.26, respectively. While metals like Cu and Pb were categorized as "no contamination," cadmium and manganese showed "considerable contamination" in sample, while Igeo values increased as Cu < Pb < Mn < Cd.
Table 4: Indicators of pollution assessment in Dry and wet seasons
Seasons | Heavy metals | CF | Igeo | PLI | ERI | EF |
Wet | Cu | 0.78 | -0.26 | 0.78 | 4 | 0.25 |
Cd | 13.8 | 0.94 | 14.11 | 241 | 3.68 | |
Pb | 1.12 | -0.11 | 1.07 | 5.6 | 0.32 | |
Mn | 3.10 | 0.37 | 2.55 | 3.42 | 1 | |
Dry | Cu | 0.82 | -0.26 | 0.80 | 4.1 | 0.24 |
Cd | 14.2 | 0.98 | 14.12 | 244 | 3.67 | |
Pb | 1.14 | -0.11 | 1.09 | 5.7 | 0.33 | |
Mn | 3.50 | 0.37 | 2.72 | 3.50 | 1 |
The degree of human effect on an area's heavy metal pollution is of concern to the EF. Metals with EF is less than one in this study, like Cu and Pb, have nothing enhancement, yet, enrichment factor less or equal to 1 and greater or equal to 3 were present in manganese and cadmium respectively, meaning they have less and moderate enrichment during the wet and dry seasons. It has been suggested that anthropogenic activities have an impact on EF>1, whereas weathering and other natural events are responsible for EF < 1.49 The analyzed sample's EF often increased as Cu244).
Level of copper in water and sediment samples
The copper quantity in both samples showed seasonal and spatial variations across the study sites. Copper (Cu) mean values in water samples from location one: 1.54 mgL-1; two: 0.5 mgL-1; and three: 0.6 mgL-1 during the dry season (Table 5). The concentrations of copper during the wet season were 1.9 milligram per liter site one, 0.64 mailgram per liter site two, and 0.62 mgL-1 site three. The values were found to range from 0.5-1.54 mgL-1 in dry weather and 0.62-1.9 mgL-1 in the rainy season. The average copper (Cu) concentrations found in most test locations in both dry and rainy seasons are below the WHO's maximum permitted levels for drinking water, but some sites, particularly in the wet season, approached this threshold, indicating potential concern for aquatic life and human use. This rise may be caused by direct or indirect input of these toxic metals from several sources, such as road runoff, agricultural discharge, watershed runoff, and rural water discharge points. Similar studies were done in Itapaji Ekiti, SW Nigeria, which is below the WHO drinking water detection level.50
During the dry season, the average copper (Cu) readings for sites one, two, and three were 2.19, 1.8, and 1.9, respectively, in the sediment sample. There was 1.95 at site three, 1.9 at two, and 2.3 at site one during the rainy season (Table 6). The average concentrations of Cu in sediment varied between 1.8 and 2.19 and 1.9 and 2.3 during dry and wet seasons, respectively. Site two exhibited the lowest concentration of Cu in the sediment in both seasons, while site one had the highest amount, similar to the water samples. The mean value of Cu in the wet season was higher than in the dry season for most test sites, which is interesting. The average copper concentrations varied between 1.8 and 2.3 mg/kg, as indicated in Table 5. Furthermore, the findings showed that the average variations in Cu contents in sediment samples from the majority of research locations were analytically considerable (p<0.05).
Cadmium level in water and sediment
Cadmium (Cd) concentration in both water and sediment samples revealed notable differences in terms of distribution, seasonal variation, and comparison with the WHO permissible limits. During dry season, the average cadmium quantity in water samples as of site one, two, and three were 0.02, 0.02, and 0.03 mgL-1, respectively. Table 5, the Cd values for the same sites were 0.01, 0.031, and 0.02 mg/L, for the period of the rainy season. Cd values were found to range from 0.02 to 0.03 mg/L during the dry season and from 0.01 to 0.031 mg/L during the wet season (Table 5). The overall mean concentration of Cd in water was 0.02±0.003 mg/L, indicating slight marginal exceedance at certain sites but generally staying within acceptable limits or remaining close to the maximum international standard of 0.03 mgL-1. The analysis of the experiment revealed that during the rainy time, the amount of cadmium was somewhat greater than during the dry season. Moreover, average value of cadmium in water samples varied depending on the sampling location and time of year. The proximity of these values to the guideline level highlights a potential risk, especially considering cadmium’s high toxicity even at low concentrations. Therefore, the statistical analysis of the study shows that the mean Cd concentrations in the lake water at several sampling sites varied significantly (P<0.05) across sites and between the two seasons.
The mean values of cadmium (Cd) concentrations in sediment samples were 0.83 mgkg-1 site one, 0.76 mgkg-1 on site two, and 0.8 mgkg-1 on site three during the dry season. Table 6 indicates that during the rainy season, the concentration on sites one, two, and three were 0.84, 0.76, and 0.83 mgkg-1, respectively. Depending on the location, the amount of Cd in the samples of sediment range starting 0.76-0.83 during the dry season and from 0.76-0.84 during the rainy season. The average cadmium levels at each sampling site ranged between 0.76 and 0.84, as shown in Table 6. The concentrations of cadmium (Cd) at every sampling location were more concentrated than the recommended limitation by WHO guidelines in sediment, which is in contrast with the findings for water samples. Table 4 shows that the average of cadmium content in the samples of the water was 0.02±0.003, which is below the WHO permissible limits, whereas in sediment samples were 0.8±0.008 (Table 6). According to these findings, although Cd levels exceeded WHO guidelines, there was a considerable variation in concentrations between sample types; in particular, during both seasons, sediment samples had greater amounts of Cd than water samples. This suggests cadmium’s strong affinity for particulate matter, making it more likely to bind to sediments and persist in the aquatic environment over time. At the Northwestern Arabian Gulf Coast, as well as Lake Naivasha, Kenya sites, cadmium concentrations were found to exceed the sediment limits set by the WHO. However, these levels were significantly lower.51,52
Table 5: Results and analysis of copper, cadmium, lead, and manganese levels in Mean ± SD in water samples in different seasons
Seasons | selected sites | detected heavy metals (mg/L) | |||
copper(Cu) | cadmium(Cd) | lead(Pb) | manganese(Mn) | ||
Dry | site one | 1.54±0.005 | 0.02±0.003 | 0.010±0.001 | 1.5±0.001 |
site two | 0.5±0.003 | 0.02±0.003 | 0.007±0.001 | 0.7±0.001 | |
site three | 0.6±0.004 | 0.03±0.002 | 0.008±0.002 | 0.71±0.003 | |
MVDS | 0.88±0.004 | 0.02±0.002 | 0.008±0.001 | 0.96±0.001 | |
Wet | site one | 1.9±0.004 | 0.01±0.004 | 0.010±0.001 | 1.6±0.002 |
site two | 0.64±0.005 | 0.031±0.003 | 0.008±0.002 | 0.7±0.001 | |
site three | 0.62±0.003 | 0.02±0.004 | 0.002±0.003 | 0.8±0.001 | |
MVWS | 1.05±0.004 | 0.02±0.003 | 0.007±0.002 | 1.03±0.001 | |
OMVDWS | 0.96±0.004 | 0.02±0.003 | 0.007±0.001 | 0.99±0.001 | |
MPL WHO | 2.0*** | 0.03 *** | 0.01*** | 0.1*** | |
SD = standard deviation of the mean, MPL=maximum permissible limit, MVDS = mean value of dry season, MVWS =mean value of rainy season, OMVDWS = overall mean value of dry and wet seasons, ***Source: WHO (2024).
The elevated levels of cadmium in sediment samples indicate that cadmium may have come from runoff that contained waste, vegetables, and irrigation water, largely because of human interest, such as the uncontrolled distribution of herbicides, small industries, chemical or soil fertilizers, and manures in the catchment area of the irrigation source. Therefore, elevated cadmium levels in sediment may be a risk factor for cadmium-related health problems. Furthermore, there may be risks associated with using this sediment for irrigation and other purposes because cadmium is becoming more widely acknowledged as a health risk in wastewater-irrigated agriculture, especially because of its associations with kidney and bone damage as well as its possible carcinogenic effects.53,54
Level of lead (Pb) in water and sediment samples
Table 5 describes the lead (Pb) concentration level in the samples of the water taken in both seasons. The mean concentration level of Pb readings was 0.010 for site one, 0.007 for site two, and 0.008 for site three in dry period. In wet term, these values increased to 0.010 at site one, 0.008 at site two, and 0.002 at site three. In water samples, lead levels remained relatively low, ranging from 0.002±0.003 to 0.010±0.001 mgL-1. The overall mean concentration was 0.007±0.001 mgL-1, which is slightly lower the WHO standard limit of 0.01 mgL-1. However, water samples from site one during both dry and wet seasons reached the maximum allowable limit, indicating site-specific concern where human and environmental exposure could pose health risks.
Table 6: Analysis of heavy metal levels in Mean ± SD in sediment samples
Seasons | selected sites | detected heavy metals (mg/kg) | |||
copper(Cu) | cadmium(Cd) | lead(Pb) | manganese(Mn) | ||
Dry | site one | 2.19±0.006 | 0.83±0.001 | 1.7±0.002 | 1.66±0.004 |
site two | 1.8±0.004 | 0.76±0.003 | 1.2±0.001 | 0.8±0.005 | |
site three | 1.9±0.003 | 0.8±0.001 | 1.8±0.001 | 0.95±0.005 | |
MVDS | 1.96±0.004 | 0.8±0.001 | 1.6±0.001 | 1.14±0.005 | |
Wet | site one | 2.3±0.003 | 0.84±0.003 | 2.2±0.008 | 1.94±0.005 |
site two | 1.9±0.003 | 0.76±0.003 | 1.2±0.001 | 0.86±0.003 | |
site three | 1.95±0.005 | 0.83±0.001 | 1.9±0.006 | 1±0.004 | |
MVWS | 2.0±0.004 | 0.81±0.002 | 1.8±0.005 | 1.3±0.004 | |
OMVDWS | 2.00±0.004 | 0.8±0.001 | 1.7±0.003 | 1.22±0.004 | |
MPL WHO | 35.7*** | 0.6*** | 35.0 *** | 1.1*** | |
SD = standard deviation of the mean, MVDS = mean value of dry season, MPL = maximum permissible limit of the World Health Organization, MVWS = mean value of wet season, OMVDWS = overall mean value of dry and wet seasons.
Although most water samples fell within safe limits, the repeated exceedance at certain sites signals the need for localized investigation and control of lead sources such as plumbing corrosion, fuel combustion, or agricultural runoff. The Pb contents in the examined water samples were equal to or less concentrated than the WHO-acceptable limits, which is comparable to the levels listed in Table 6.
Lead (Pb) levels in sediment samples from this study were 1.7 at site one, 1.2 at site two, and 1.8 at site three during the dry season. The levels increased to 2.2 at site one, remained at 1.2 at site two, and reached 1.9 at site three during the wet season (see Table 6). Site one had the highest Pb level, and site two had the lowest during the dry season. The mean concentration of lead in sediment samples in the dry season was less than in the wet one, ranging from 1.2 to 1.8 and from 1.2 to 2.2, respectively. Pb concentrations at all sediment monitoring sites typically fell between 1.2 and 2.2 mg/kg. Overall mean values of the lead in sediment and water samples were 1.7±0.003 and 0.007±0.001, respectively, based on the investigations shown in Tables 5 and 6. Therefore, sediment samples had Pb levels that were significantly (p<0.05) below the recommended limits. This indicates that while a lead does accumulate in sediments, the levels are not yet at a critical threshold. The slightly higher concentrations observed during the wet season, especially at site one, and may be attributed to surface runoff transporting lead from surrounding land areas into the aquatic system. Similar reports of the Pb levels in Dianchi basin, China, and Thailand, which were within and below the WHO's maximum detection standards.55,56 The presence of lead in both water and sediment, albeit within permissible limits, suggests ongoing low-level contamination likely from diffuse or non-point sources. Lead concentrations in aquatic systems, which they attributed to precipitation, street runoff, industrial discharges, lead-containing pesticides, and municipal wastewater.57
Level of manganese in water and sediment samples
The analysis of the manganese (Mn) in both water and sediment samples reveals a significant level of contamination, particularly in water, with seasonal and spatial variations. The average values of Mn in this study were 1.5 at site one, 0.7 at site two, and 0.71 at site three during the dry season in the water sample. The measurements recorded were 1.6 at site one, 0.7 at site two, and 0.8 at site three during the wet season (refer to Table 5). The average Mn concentrations started from 0.7-1.5 in the period of the dry & from 0.7-1.6 throughout the rainy period. Site two had the lowest Mn concentrations in both seasons, while site one had the highest. The findings showed that in the wet period, Mn amount were greater than in dry time. The overall mean value for manganese in the water sample was 0.99±0.001 mgL-1, which is nearly ten times higher than the fresh water limit. This persistent exceedance across all sites and seasons indicates widespread contamination, possibly resulting from natural leaching of manganese-rich geological formations, erosion, or anthropogenic activities such as agricultural runoff, industrial effluents, or untreated wastewater discharge. According to this study, Lake Haramaya’s Mn levels may be harmful for human consumption because they are marginally higher than usual. The health effects of increasing exposure become substantial when water concentrations are above the WHO drinking water guidelines. Manganese is one of the important heavy metals that the human body needs for certain processes, but excessive exposure can cause heavy metal poisoning symptoms. Particularly when consumed over a long period, elevated manganese concentrations can be extremely harmful to one's health and contribute to several chronic illnesses.58,59 Similar research found that Mn concentrations in Delta Lake, Egypt, and Bangladesh, started from 0.5 to 1.45, beyond the maximum limit recommended for drinking water.60,61
The mean values of manganese (Mn) at site one, site two, and site three were 1.66±0.04, 0.8±0.005, and 0.95±0.005, respectively, throughout the dry season in sediment samples. Site two had the lowest concentration during this season, whereas site one had the highest. As shown in Table 6, the Mn concentrations at site one, site two, and site three during the wet season were 1.94±0.005, 0.86±0.003, and 1±0.004, respectively. Once more, site two had the lowest concentration, whereas site one had the highest. Overall, in the wet period, the average Mn level in sediment samples range started 0.86-1.94, while in dry period; from 0.8-1.66. Therefore, in sediment samples, manganese concentrations were also elevated but generally closer to the WHO sediment guideline of 1.1 mg/kg. The average values started from 0.8±0.005-1.94±0.005 mgkg-1, with the highest levels found at site one during the wet season. The overall mean manganese concentration in sediment was 1.22±0.004 mg/kg, indicating a slight exceedance of the permissible limit. The higher manganese levels in the wet season suggest increased input from surface runoff and erosion processes during rainfall, contributing to the accumulation in bottom sediments. A similar level of Mn concentration was found at Eawag, the Swiss Federal Institute of Aquatic Science and Technology, China, in freshwater sediment.62,63 As indicated in Tables 5 and 6, the average Mn concentrations in sediment and water samples were 0.8 to 1.94 and 0.7 to 1.6, respectively, across all sampling sites. Based on the analysis, manganese concentration both in water and sediment samples consistently exceeds WHO limits, particularly during the wet season, indicating a significant source of contamination. This suggests a potential risk to human health, especially where water is used for consumption or agricultural process, and highlights the require for immediate management techniques and regular monitoring.
Discussion
The mean values with standard deviations for key physico-chemical analysis, temperature, pH, electrical conductivity (EC), and total dissolved solids (TDS) of water samples collected from three sites during both dry and wet seasons. The range of temperature started from 24-27°C, with the overall mean being 25.5±0.004°C, which is slightly below the dry season, but still acceptable within the WHO permissible range of 25-30°C. The pH values were stable across all sites and seasons, ranging narrowly between 7.11 and 7.15, indicating neutral to slightly basic water that falls well in standard range of 6.5 to 8.5. The electrical conductivity (EC) results presented reflect the concentration of dissolved ions in water at different sites and seasons, which were varied slightly, from 1.28 to 1.55 mS/cm, equivalent to 1280-1550 µS/cm. These values are all within the WHO/ESEPA permissible range of 500-1500 µS/cm, except site one in the dry season slightly exceeds the upper limit, indicating elevated ion concentration, possibly due to evaporation, and reduced water volume. TDS concentration ranged between 541 and 570 mg/L, with an overall mean of 557 ± 0.004 mg/L, also comfortably below the 1000 mg/L limit set by WHO.37,38 Overall, the findings show that there is little seasonal change in the water’s physico-chemical characteristics, which are within safe and acceptable bounds for drinking and household usage.
The heavy metals analysis showed the amount of copper, cadmium, lead, and manganese in water and sediment samples taken from three various locations during both dry & wet seasons. Copper concentrations ranged from 0.5 mg/L at site two in the dry period to 1.9 mgL-1 at site one in the wet season. The seasonal mean values were 0.88 mgL-1 in the dry time and 1.05 mgL-1 in the wet period, with an overall mean of 0.96 mgL-1, all of which are under WHO maximum permissible boundary. In the sediment sample, copper levels ranges started from 1.8-2.3 mgkg-1 & were well below the permissible limits. The concentrations of cadmium were fairly consistent in the water sample, with an overall mean of 0.02 mgL-1, which is within the WHO limit of 0.03 mg/L. In contrast, the cadmium concentrations in the sediment sample were relatively consistent across seasons, ranging between 0.76 and 0.84 mgkg-1, with an overall mean of 0.8 mgkg-1, which exceeds the WHO limit of 0.6 mg/kg, indicating potential contamination. The increase in cadmium levels in sediment samples of Haramaya Lake is primarily linked to human activities in the surrounding areas. Agricultural runoff is the major source, as phosphate fertilizers widely used in nearby farms often contain cadmium as an impurity. During rainfall, surface runoff carries these cadmium-laden particles into the lake, where they eventually settle in the sediment. It tends to bind strongly with fine particles such as clay and organic matter present in the sediment, making it persistent and less likely to be removed through natural processes.64-66 Furthermore, untreated domestic sewage and wastewater from nearby towns may contain cadmium from household products such as batteries, plastics, and detergents. These wastes enter the lake through open drainage systems and contribute to the heavy metal load in the sediments. The lead concentrations found between 0.002 & 0.010 mgL-1, & 1.2 & 2.2 mgL-1, respectively, in water and sediment samples. The overall mean of lead concentration was 0.007 mgL-1 in the water sample and 1.7 mg/kg in sediment, remaining safely below the WHO guideline. The concentrations of manganese increased in the wet period compared to dry period, and the overall mean of 1.22 mg/kg slightly surpassed the WHO limit of 1.1 mg/kg. The increase in manganese concentration in both the water and sediment of Haramaya Lake can be attributed to a combination of natural geochemical processes and human activities in the surrounding area. Naturally, manganese is released into the water from the weathering of rocks and soil erosion in the watershed.67,68 Human activities have further intensified manganese accumulation.69,70 Agricultural runoff contains fertilizers and pesticides, along with wastewater discharge from nearby settlements, may introduce additional manganese in the lake system and bind easily to fine sediment particles like clay and organic matter, making it prone to accumulate in sediments over time. Similar results were found, indicating that cadmium sediment levels in the mouth of the Sher Karuturi, Flamingo, and Malewa Rivers were higher than the WHO criteria for Kenyan sediment quality.65,71 Higher levels of some heavy metals suggest that it acts as the last storage location in aquatic environments.72,73 This implies that the use of polluted silt for agricultural irrigation through drainage systems may be because of the buildup of elevated cadmium and manganese levels at most test sites, endangering aquatic and terrestrial creatures such as fish, plants, and people.
To mitigate the environmental and economic risks associated with heavy metal contamination in Lake Haramaya, a series of policy and management interventions is recommended. First, it is crucial to strengthen regulatory monitoring by establishing a regular and systematic assessment of heavy metal concentrations in both water and sediments, coordinated by national or regional environmental agencies. This will enable early detection of pollution trends and timely interventions. Secondly, controlling both point and non-point pollution sources is essential. This involves enforcing strict regulations on agricultural runoff, domestic sewage discharge, and industrial waste disposal in areas surrounding the lake. Additionally, raising community awareness through educational programs targeting local farmers and residents can foster responsible environmental behavior and encourage proper waste management practices. Developing accessible and cost-effective water treatment solutions is also important to ensure safe water for drinking and agricultural use among nearby communities. Promoting sustainable land use practices like eco-friendly farming techniques and the establishment of vegetative buffer zones can significantly reduce pollutant inflow in to the lake. Finally, the findings of this study should be integrated into local and regional development plans to ensure that future economic growth aligns with environmental conservation goals. These measures, if effectively implemented, will contribute to the sustainable management and restoration of Lake Haramaya’s ecological integrity.
Conclusions
The quantities of the chosen heavy metals, such as Cu, Cd, Pb, and Mn in sediment and water samples from Lake Haramaya, Ethiopia, were examined in this study. The amounts of particular heavy metals in all selected samples varied by sampling site and season, as did the physicochemical characteristics of water. The majority of the findings from the analysis of the physicochemical properties of Haramaya Lake's water and sediment fell within or below the WHO's recommended values required for aquatic creatures' survival, metabolism, and physiology, except for electrical conductivity, which is slightly exceeded in site one during the dry season. The major findings reveal that the assessment of copper, cadmium, lead, and manganese levels in both water and sediment samples shows varying degrees of contamination and potential risks. Copper concentrations in both water and sediment were consistently within WHO permissible limits, indicating no immediate environmental or health concern from copper at the studied sites. Cadmium, while within safe limits in water, exceeded the WHO guideline in sediment, suggesting long-term accumulation and potential ecological risks, particularly to benthic organisms. Lead concentrations remained below the WHO permissible limits both in sediment and water samples. In contrast, manganese levels were significantly above WHO permissible limits in both water and sediment, indicating persistent contamination and potential health threats, particularly if the water is used for drinking or irrigation. The study also found spatial and seasonal variations in metal levels, with higher concentrations typically observed during the dry season. Despite these critical insights, the study had several limitations. Sampling was limited to four sites and two seasons, which may not capture the full spatial and temporal variability of the lake system. In addition, only a selected number of metals were analyzed, excluding others such as mercury, arsenic, or chromium that may also be of concern.
To address the environmental and public health concerns arising from contamination in freshwater and sediment systems, local authorities should consider implementing regular biomonitoring programs using bioindicator species such as fish and benthic invertebrates. These organisms can help detect the bioaccumulation of harmful substances and serve as early warning systems for ecological deterioration. In addition, stricter regulations on industrial and municipal wastewater discharge should be enforced, particularly targeting pollutants like copper, manganese, lead, and cadmium. Regular monitoring and compliance checks can ensure adherence to safe discharge limits. Authorities should also promote community awareness about the sources and impacts of water pollution, encouraging active participation from local stakeholders in decision-making processes. Lastly, it is essential to develop localized sediment quality guidelines aligned with international standards like WHO and USEPA to support effective sediment management and protect aquatic ecosystems. Together, these measures can significantly enhance environmental governance and safeguard both ecosystem and human health.
Acknowledgement
Authors express heartfelt appreciation and thanks to the Department of Biology and Biotechnology, Haramaya University, Ethiopia for providing funding and infrastructural facilities to conduct the experimental studies. Authors are also thankful to the Department of Biotechnology and Food Technology, Punjabi University, Patiala, India for necessary support and library facilities. Authors also present special gratitude to Meseret Desalegn for her unreserved advice and constructive suggestions throughout this study.
Funding Sources
Authors (Yaecob Gebre Leleko (YGL), Sissay Menkir (SM), Endale Teju (ET) express heartfelt appreciation and thanks to Department of Biology and Biotechnology, Haramaya University, Ethiopia, for providing funding and infrastructural facilities to conduct the experimental studies (HURG-2017-05-02-06).
The authors do not have any conflict of interest
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Permission to reproduce material from other soures
Not Applicable
Author Contributions
Yaecob Gebre Leleko: Date collection, conducted experiments, analysis, and wrote the original draft of the research work.
Sissay Menkir: Funding acquisition for research project, providing resources, and supervising the whole research project works.
Endale Teju: Designed the project and visualized experimental works.
Ranjeeta Bhari: Review and data analyzed.
Balwinder Singh Sooch: Analyzed the research data, edited and proofread the manuscript.
References
- Kilic Z. The importance of water and conscious use of water. Int J of Hydro. 2020; 4(5):239-241.
CrossRef - Ahmed A.A, Oyedele L, Sayed S, Abdoulhalik A, Moutari S. Applications of machine learning to water resources management: A review of present status and future opportunities. J of Clea Prod. 2024; 441: 1-18.
CrossRef - Musie W, and Gonfa G. Fresh water resource, scarcity, water salinity challenges and possible remedies: A review. Heliyon. 2023; 9(8): 1-18.
CrossRef - Su Q, Kambale R.D, Tzeng J.H, Amy J.L, Ladner D.A, Karthikeyan R. The growing trend of saltwater intrusion and its impact on coastal agriculture: challenge and opportunities. Sci of Tot Environ. 2025; 966: 1-15
CrossRef - Zhao D, Liu J, Sun L, Hubacek, H, Pfister S, Feng K, Zheng H, Peng X, Wang D, Yang H, Shen L, Lunm F, Zhao X, Bin Chen B, Keskinen M, Zhang S, Cai J, Varis O. Water consumption and biodiversity: Responses to global emergency events. Sci Bullet. 2024; 69:2632-2646.
CrossRef - Vanham D, Alfieri L, Flörke M, Grimaldi S, Lorini V, Roo A, and Feyen L. The number of people exposed to water stress in relation to how much water is reserved for the environment: a global modeling study. The Lan Plane Heal. 2021; 5(11): 766-774.
CrossRef - Ahmed S.F, Kumar P.S, Kabir M, Zuhara F.T, Mehjabin A, Tasannum N, Hoang, A.T, Kbir Z, and Mofijur M. Threats, challenges and sustainable conservation strategies for freshwater biodiversity. Environ Res. 2022; 214(1):113808.
CrossRef - Fedele G, Donatti C.I, Bornacelly I, Hole D.G. Nature-dependent people: Mapping human direct use of nature for basic needs across the tropics. Glo Environ chan. 2021; 71: 1-9.
CrossRef - Plessis A. Persistent degradation: Global water quality challenges and required actions. One Eart. 2022; 5(2): 129-131.
CrossRef - Akhtar N, Izzuddin M.S.I, Ahmad S.B, and Umar K. Various natural and anthropogenic factors are responsible for water quality degradation. Water Qual and Pub Heal. 2021; 13(19): 2660.
CrossRef - Hasan M.F, Alam M.N, Salam M.A, Rahman H, Paul S.C, Rak A.E, Ambade B, and Towfiqul R.M. Health risk and water quality assessment of surface water in an urban river of Bangladesh. Susta. 2021; 13(12):6832.
CrossRef - Malik N, Biswas A.K, Qureshi T.A, Borana K. and Virha R. Bioaccumulation of heavy metals in fish tissues of a freshwater lake of Bhopal. Environ Monit Assess. 2020; 160: 267-267.
CrossRef - Service T, Cassidy R, Atcheson K, Farrow L, Harrison T, Jack P, and Jordan P. A national-scale high-resolution runoff risk and channel network mapping workflow for diffuse pollution management. J of Environ Manag. 2024; 368: 122110.
CrossRef - Xia Y, Zhang M, Tsang D.W, Geng N, Lu D, Zhu L, Igalavithana A.D, Dissanayake P.D. Rinklebe J, Yang X, and Ok Y.S. Recent advances in control technologies for non-point source pollution with nitrogen and phosphorous from agricultural runoff: current practices and future prospects. Appl Biol Chem. 2020; 63(8):1-13.
CrossRef - Baral D, Bhattarai A, Chaudhary N.K. Aquifer pollution by metal-antibiotic complexes: Origins, transport dynamics, and ecological impacts. Eco Toxic and Environ Saf. 2024; 288: 1-14.
CrossRef - Rajasimman P.S.M, Saravanan V, Baskar G, Rajeshkannan R, and Arnica G, Pugazhendhi A. Comprehensive review on toxic heavy metals in the aquatic system: sources, identification, treatment strategies, and health risk assessment. Environ Rese. 2024; 258: 1-20.
CrossRef - Briffa J, Sinagra E, and Blundell R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliy. 2020; 6(9): 1-20.
CrossRef - Membere E, and Abdulwasiu M. Heavy metals concentration in water, sediment, and fish around escravos river, Nigeria. Wor J of Rese and Rev. 2020; 10(2):28-37.
- Rasin P, Ashwathi A.V, Basheer S.M, Garrote C.A, Haribabu J, Santibanez J.F, Arulraj A, and Mangalaraja R.V. Exposure to cadmium and its impacts on human health: A short review. J of Haz Mate Adv. 2025; 17: 1-12.
CrossRef - Yang T, Lee S.Y, Park K.C, Park S.H, Chung J, and Lee S. The effects of selenium on bone health: from element to therapeutics. Molecu. 2022; 27(2):392.
CrossRef - Belachew M.M and Demse B.G. The current irrigation potential and irrigated land in Ethiopia-A review. Int J of Cur Res and Aca Rev. 2022; 10(01): 64-72.
- Berhe H, and Bezabeh E. Implications of environmental pollutions on Ethiopian fisheries: review study. Rese on Hum and Soc Sci. 2021; 11(17):1-12.
- Saravanan P, Rajasimman M, Saravanan V, Baskar G, Rajeshkannan R, and Arnica G, Pugazhendhi A. Comprehensive review on toxic heavy metals in the aquatic system: sources, identification, treatment strategies, and health risk assessment. Environ Rese. 2024; 258: 1-20.
CrossRef - Ullah M.I.S, Shahriara S, Naidua R, Rahman M.A. Concentrations of potentially toxic and essential trace elements in marketed rice of Bangladesh: Exposure and health risks. J of Food Comp and Anal. 2023; 117: 1-13.
CrossRef - Mitra S, Chakraborty A.J, Tareq A.M, Emran T.B, Nainu F, Khusro A, Idris A.M, Khandaker M.U, Osman H, Alhumaydhi F.A, and Gandara J.S. Impact of heavy metals on the environment and human health: Novel therapeutic insights to counter the toxicity. J of Kin Sau Unive Sci. 2022; 34(3):101865.
CrossRef - Wu H, Zhi Y, Zhang Y, Xiao X, Yu F, China P.R, Cao G, Xu X. Source-oriented health risk of heavy metals in sediments: A case study of an industrial city in China. Ecotox and Environ Saf. 2025; 292: 1-11.
CrossRef - Haile E., R.S, Reddy U, and Awol A.Y. The drivers for the collapse of Lake Haramaya and proposed integrated rehabilitation strategies. Inter Associ for Hydro-Environ Engin and Rese. 2022; 21(9): 1-39.
- Choudhury B.U, Nengzouzam G, and Islam A. Evaluation of climate change impact on soil erosion in the integrated farming system based hilly micro-watersheds using Revised Universal Soil Loss Equation. Catena. 2022; 214: 1-11.
CrossRef - Seleshi B.A, Aster D.Y, Makonnen L, Willibald L, Mekonnen A, and Tena A. Water resource and irrigation in Ethiopia. Int Water Manag Inst. 2015; 1-83.
- Hasana M.A, Satter M.A, Da A.K, and Asif M. Detection of heavy metals in fish muscles of selected local fish varieties of the Shitalakshya River and probabilistic health risk assessment. Meas of Food. 2022; 8: 100065.
CrossRef - Dange S, Arumugam K, Vijayaraghavalu S.S. Geochemical insights into heavy metal contamination and health hazards in Palar River Basin: A pathway to sustainable solutions. Ecol Indic. 2024; 166: 1-15.
CrossRef - WHO guidelines for drinking-water quality, 4th edition (incorporating the 1st and 2nd addenda, (published May 2023).
- FAO Guidelines for Trace Elements in Irrigation Water and the state of food security and nutrition in the world (2024).
- United States environmental protection agency, freshwater sediment screening benchmarks, region 3 risk assessment guidance (last updated September 6, 2024).
- World health organization (WHO) guide line for drinking water quality 2024. https://iris.who.int/bitstream/handle/10665/375822/9789240088740-eng.pdf.
- Teferi T.D, and Gezahagn T.G. Determination of metals in water samples within the irrigation area in Telo District, Kaffa Zone, and South Western Ethiopia. Heliy. 2024; 10(7): e29003.
CrossRef - Kothari V, Vij S, Sharma S.K, Gupta N. Correlation of various water quality parameters and water quality index of districts of Uttarakhand. Environ and Sust Indic. 2021; 9: 1-8.
CrossRef - Shaikh S. Estimation of water quality physico-chemical parameters of drinking water sources of Usroli village of Murud Tehsil district- Raigad, Maharashtra, India. Scho Rese J for Interdis Stu. 2025; 7(39): 21-27.
- Ngyah K, and Bih T. Physicochemical analysis of potable water in baham community. Glob Sci J. 2021; 9(11):1119-1140.
- Risal S.Y, Islam S, Ferdous J, Siddik M.N.A, and Bakshi P.K. Natural radioactivity and heavy metal contamination in edible fish, shellfish and mollusks at the Bay of Bengal, Kuakata, Bangladesh. Heliy. 2024; 10: 1-16.
CrossRef - Kiruba T, Jayaprakash P, Gandhi M.S, Juliet J, Joy J, Ravisankar R, Venkatamuthukumar J. Assessment of metal contamination in sediment of Kerala Coast, India using EDXRF technique and Multivariate Statistical method. Nucl Anal. 2025; 4(1): 1-13.
CrossRef - Aja D, Okolo C.C, Nwite N.J, and Njoku C. Environmental risk assessment in selected dumpsite in Abakaliki metropolis, Ebonyi state, Southeastern, Nigeria. Environ Chall. 2021; 4: 1-9.
CrossRef - Muzeregni C. Enrichment and Geoaccumulation of Pb, Zn, As, Cd and Cr in soils near New Union Gold Mine, Limpopo Province of South Africa. Int Min Water Assoc Congr. 2017; 25(30): 720-727.
- Afolabi O.O, Wali E, Ihunda E.C, Orji M.C, Emelu V.O, Bosco-Abiahu L.C, Ogbuehi N.C, Asomaku S.O, Wali O.A. Potential environmental pollution and human health risk assessment due to leachate contamination of groundwater from anthropogenic impacted site. Environ Chall. 2021; 9: 1-10.
CrossRef - Orcan F. Parametric or non-parametric: Skewness to test normality for mean comparison. Int J of Assess Tool in Educ. 2020; 7(2):255-265.
CrossRef - Shah A, Arjunan A, Baroutaji A, Zakharova J. Areview of physicochemical and biological contaminants in drinking water and their impacts on human health. Water Sci and Engin. 2023: 16(4): 333-344.
CrossRef - Alsafran M, Usman K, AlJabri H, and Rizwa M. Ecological and health risks assessment of potentially toxic metals and metalloids contaminants: a case study of agricultural soils in Qatar. Toxics. 2021; 9 (35):1-15.
CrossRef - Ohiagu F.O, Lele K.C, Chikezie P.C, Verla A.W, and Enyoh C.E. Pollution profile and ecological risk assessment of heavy metals from dumpsites in Onne, Rivers state, Nigeria. Chem Afr. 2020; 4: 207-216.
CrossRef - Sepúlveda L, Pasquini A, Temporetti P, Lecomte K. Unraveling the sources of major, trace and rare earth elements in the waters of a high latitude proglacial environment: Weathering vs. atmospheric signature in Northern Patagonia. Chem Geol. 2024; 646: 1-13.
CrossRef - Abdu-Raheem Y.A, Oyebamiji A.O, Afolagboye L.O, Talabi A.O. Assessment of ecological and health risk impact of heavy metals contamination in stream sediments in Itapaji-Ekiti, SW Nigeria. J of Tra Ele and Mine. 2024; 8: 100121.
CrossRef - Alharbi T, Nour H.E, El-Sorogy A.S, Al-Kahtany K, and Zumlot T. Health risk assessment and contamination of lead and cadmium levels in sediments of the northwestern Arabian Gulf coast. Heliy. 2024; 10(16): 1-12.
CrossRef - Olando G, Olaka L.A, Okinda P.O, and Abuom P. Heavy metals in surface sediments of Lake Naivasha, Kenya: spatial distribution, source identification and ecological risk assessment. SN App Sci. 2020; 279(2): 1-1-14.
CrossRef - Alengebawy A, Abdelkhalek S, Qureshi S, and Wang M. Heavy metals and pesticides toxicity in agricultural soil and plants: ecological risks and human health implications. Natio Lib of Medi. 2021; 9(3):42.
CrossRef - Decharat S, and Pan-in P. Risk assessment of lead and cadmium in drinking water for school use in nakhon province, Thailand. Environ Anal Heal and Toxic. 2020. 35(1):1-6.
CrossRef - Liang H.Y, Ding T.T, Zhang Y.H, et al., Heavy metals in sediments of the river-lake system in the Dianchi basin, China: Their pollution, sources, and risks. Sci of the Tot Environ. 2024; 957: 1-12.
CrossRef - Kurwadkar S, Sethi S.S, Mishra P, and Ambade B. Unregulated discharge of wastewater in the Mahanadi River Basin: Risk evaluation due to occurrence of polycyclic aromatic hydrocarbon in surface water and sediments. Marr Pollu Bull. 2022; 179: 1-10.
CrossRef - Alia M.M, Alia M.L, Islam M.S, Rahman M.D. Preliminary assessment of heavy metals in water and sediment of Karnaphuli River, Bangladesh. Environ Nanotech, Monit and Mang. 2023; 5: 27-35.
CrossRef - Huang Q, Wan J, Nan W, Li S, He B, and Peng Z. Association between manganese exposure in heavy metals mixtures and the prevalence of sarcopenia in US adults from NHANES 2011–2018. J of Haz Mater. 2024; 464: 15: 133005.
CrossRef - Rushdi M.I, Basak R, Das P, Ahamed T, and Bhattacharjee S. Assessing the health risks associated with elevated manganese and iron in groundwater in Sreemangal and Moulvibazar Sadar, Bangladesh. J of Haz Matt Adv. 2023; 10: 1-10.
CrossRef - Uddin M.J, and Jeong Y.K. Urban river pollution in Bangladesh during last 40 years: potential public health and ecological risk, present policy, and future prospects toward smart water management. Heliyon. 2021; https://doi.org/10.1016/j.heliyon.2021.e06107.
CrossRef - Hu X, Tlili A, Schirmer K, Eawag M.B, Swiss Helmut Bürgmann S.H. Metal concentration in freshwater sediments is linked to microbial biodiversity and community composition. Environ Inter. 2025; 199: 1-13.
CrossRef - Rahman M.S, Ahmed Z, Seefat S.M, Alam R, Islam A.R, Choudhury T.R, Begum B.A, Idris A.M. Assessment of heavy metal contamination in sediment at the newly established tannery industrial Estate in Bangladesh: A case study. Environ Chem and Ecotox. 2022; 4: 1-12.
CrossRef - Oladimeji T.E, Oyedemi M, Emetere M.E, Odunlami O.A, Agboola O, Adeoye J.B. Review on the impact of heavy metals from industrial wastewater effluent and removal technologies. Heliyon. 2024; 10(23): 1-30.
CrossRef - Li Q, Li L, Yin B, Lin X, Xiao A , Xue W, Liu H, Li Y. Accumulation and distribution of cadmium at organic-mineral micro-interfaces across soil aggregates. Ecotox and Environ Saf. 2025; 289(1): 1-13.
CrossRef - Xu W, Tan D, Liu C, Gao T, Zhu J.M, Liu Y, Bu H, Tong H, Chen M. Adsorption of cadmium on clay-organic associations in different pH solutions: The effect of amphoteric organic matter. Ecotox and Environ Saf. 2022; 236(1): 1-10.
CrossRef - Qu S, Wu W, Nel W, and Ji J. The behavior of metals or metalloids during natural weathering: A systematic study of the mono-lithological watersheds in the upper Pearl River Basin, China. Sci of the Tot Environ. 2020; 708: 1-10.
CrossRef - Zhang Z, Zhu Q, Yang P, Zhu M, Wen K, Mao H, Zhao Z, and Liu C. Manganese oxidation states and availability in forest weathering profiles of contrasting climate. Geochem et Cosmochi Acta. 2025; 388: 221-235.
CrossRef - Yu X, Zhou Q, Huang X.Y, Chen M, Zhou J, Zhou T, Hu P, Pane X, Cheng M, Y Luo Y, Christie P, Wu L. Elucidating the effects of manganese on the growth and cadmium accumulation of OsNRAMP5 mutant rice. Ecotox and Environ Saf. 2025; 291: 1-9.
CrossRef - Dey S, Tripathy B, Santosh M, Das A.P. Ecotoxicological consequences of manganese mining pollutants and their biological remediation. Environ Chem and Ecotox. 2023; 5: 55-61.
CrossRef - Hussain M, Ali A.S, Haruna A, Kousar T, Zango Z.U, Mahmood F, Adamu H, Mohammed G, Ibrahim K, Abdulganiyyu A, Keshta B.E. Efficient removal of manganese (II) ions from aqueous solution using biosorbent derived from rice husk. Sust Chem One Wor. 2025; 5: 100047.
CrossRef - Preonty N.E.J, Hassan M.N, Mahim M.A, Selim A.H.M, Bangladesh R, Ishtiak M, Rasel A. Pollution and health risk assessment of heavy metals in surface water of the industrial region in Gazipur, Bangladesh. Environ Chem and Ecotox. 2025; 7: 527-538.
CrossRef - Ray S, and Vashishth R. From water to plate: Reviewing the bioaccumulation of heavy metals in fish and unraveling human health risks in the food chain. Emer Conta. 2024; 10: 1-14.
CrossRef - Kakade A, Sharma M, Salama E.S, Zhang P, Zhang L, Zing X, Yue J, Song Z, Nan L, Yujun S, and Li X. Heavy metals (HMs) pollution in the aquatic environment: Role of probiotics and gut microbiota in HMs remediation. Environ Rese. 2023; 223(15): 115186.
CrossRef






