Integrated Evaluation of Toxicity and Seasonal Water Productivity in Urban Lakes: A Multimodal Approach to Aquatic Ecosystem Health
1
Department of Microbiology and Botany,
School of Sciences,
JAIN (Deemed to be University),
Bangalore,
Karnataka
India
2
Department of Forensic Sciences,
School of Sciences,
JAIN (Deemed to be University),
Bangalore,
Karnataka
India
Corresponding author Email: pr.mathews@jainuniversity.ac.in
DOI: http://dx.doi.org/10.12944/CWE.20.2.9
Copy the following to cite this article:
Raj M. P, Philip R. S. Integrated Evaluation of Toxicity and Seasonal Water Productivity in Urban Lakes: A Multimodal Approach to Aquatic Ecosystem Health. Curr World Environ 2025;20(2). DOI:http://dx.doi.org/10.12944/CWE.20.2.9
Copy the following to cite this URL:
Raj M. P, Philip R. S. Integrated Evaluation of Toxicity and Seasonal Water Productivity in Urban Lakes: A Multimodal Approach to Aquatic Ecosystem Health. Curr World Environ 2025;20(2).
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2025-06-30 |
|---|---|
| Accepted: | 2025-07-18 |
| Reviewed by: | 
 Jayvardhan Balkhande
Jayvardhan Balkhande
|
| Second Review by: |

 Haeruddin Daeng Mile
Haeruddin Daeng Mile
|
| Final Approval by: | Dr. R K Aggarwal |
Introduction
Urban lakes are ecosystems playing a vital role in groundwater recharge and microclimate regulation.1 Encroachment of urban lands has led to surface sedimentation, pesticide and untreated sewage disposal into lakes, causing oxygen depletion, eutrophication and biodiversity loss.2 Globally, lakes in cities like Nairobi, Beijing, Sao Paulo, etc have exhibited drastic anthropogenic stress, impairing aquatic productivity and ecosystem function.3
Urbanization has greatly affected the ecological condition of aquatic systems, particularly in fast-growing cities like Bangalore, India. Urban lakes, which were once crucial for biodiversity and water regulation, now struggle with intense human-induced pressures such as sewage pollution, solid waste accumulation, and eutrophication.4, 5 These issues have led to poorer water quality, decreased aquatic life, and disrupted ecosystems.6
Primary productivity, an essential measure of aquatic health, indicates a water body's capacity for autotrophy and is greatly affected by factors like temperature, nutrient levels, light penetration, and pollution.7, 8 Metrics such as chlorophyll-a concentration and oxygen production via photosynthesis offer insights into trophic status and ecological balance.9 Seasonal changes, especially during the monsoon, affect nutrient cycling and influence productivity dynamics.10 Besides physicochemical indicators, biological assays with model organisms like zebrafish provide sensitive and comprehensive tools for ecological monitoring.11 Zebrafish embryos and adults have been used to detect aquatic toxicity, developmental abnormalities, and neurotoxicity through parameters such as heartbeat, somite development, and acetylcholinesterase (AChE) activity.12,13 AChE inhibition, in particular, is a recognized biomarker for exposure to neurotoxic substances such as organophosphates and heavy metals.14 Despite the ecological importance of Bangalore’s urban lakes, few studies have combined limnological and toxicological methods to evaluate their ecological health. This research aims to assess the seasonal productivity of three urban lakes viz. Ullal, Herohalli, and Lingadheeranahalli—through a combination of seasonal productivity analysis and zebrafish-based toxicity tests, providing a multimodal framework for monitoring urban lake health. Previous studies on these lakes showed varying limnological characteristics.15
Materials and Methods
Study area
 | Figure 1: Sampling stations along with the coordinates for the three lakes
|
The current research was conducted on Ullal Lake (12.9609° N, 77.4807° E), Herohalli Lake (12.9891° N, 77.4896° E), and Lingadheeranahalli Lake (13.0035° N, 77.4827° E) in the northern urban district of Bangalore as depicted in Figure 1. All three lakes are situated in residential areas and have faced considerable human-induced stress due to urbanization.
Limnological Assessments
The research examined three sampling stations from each lake: open, deposit, and inlet stations. The assessments concentrated on water productivity by measuring oxygen-dependent parameters, specifically primary productivity and chlorophyll.
Primary Productivity
Light and Dark bottle method
Water samples collected from three sampling stations were transferred into BOD bottles; one was kept in dark conditions, and the other was exposed to light conditions for 24 hours. Initial and post-incubation DO was measured using Winkler’s method and a calibrated DO sensor (Lutron DO-5509).16 The net productivity, respiration rate and gross productivity were calculated using the following formulae.
Respiration Rate (R) (mg O2/L/day)
R = (Initial oxygen Concentration (CO) – Oxygen concentration dark (CD) / Time (t)
Gross Primary Productivity (PG) (mg O2/L/day)
PG = (Oxygen concentration light (CL) - Oxygen concentration dark CD) / Time (t)
Net Primary Productivity (PN) (mg O2/L/day)
PN = PG - R
Chlorophyll method
The lake water samples collected were filtered using Whatman filter paper (Millipore grade 1), which was then finely cut into small pieces along with 80% acetone. The samples were centrifuged (4500 rpm, 10 minutes), and the supernatant was collected. Absorbance was measured using a Labman LMSP UV 1200 spectrophotometer at 645 nm, 663 nm, and 652 nm for chlorophyll a, chlorophyll b, and total chlorophyll, respectively, using acetone as a blank solution. The absorbance is then used in Arnon’s equation.17
Chlorophyll a (mg/L) = 12.7 × A663 - 2.69 × A645
Chlorophyll b (mg/L) = 22.9 × A645 - 4.68 × A663
Total Chlorophyll (a + b, mg/L) = 20.2 × A645 + 8.02 × A663
Zebrafish culture
Adult D. rerio (Hamilton,1822) weighing between 0.85g and 1.50g were grown in well-aerated aquarium tanks (5 liter capacity) regulated at 25°C - 30°C with a 13-hr light and 11-hr dark cycle. For spawning, which was induced during the morning hours, one male fish and three female fish were taken into small breeding tanks fitted with a mesh, as demonstrated by Rahman et al.13
Somite stage and Heartbeat assessment
Harvested zebrafish embryos were transferred into small tanks containing reverse osmosis (RO) water and individual lake water samples. Developmental abnormalities were evaluated through microscopic examination. 12 Heart rate measurements were taken during the torpedo stage.11 The experimental setup was adopted from Brand and Granato.18
Acetylcholine esterase activity
Nine adult male zebrafish (three per group) were obtained and exposed to lake water samples under controlled conditions for 24 hours. After exposure, the fish were euthanized, and their brain tissues were dissected and homogenized in 0.1 M phosphate buffer (pH 7.4). The homogenate was centrifuged at 4°C at 10,000 rpm for 10 minutes, and the resulting supernatant served as an enzyme source for acetylcholinesterase (AChE) using Ellman’s method which was slightly modified.19 Acetylthiocholine iodide (ATCI) acted as the substrate, while Ellman’s reagent, 5,5'-dithiobis-2-nitrobenzoic acid (DTNB), functioned as the chromogen. A reaction mixture of 300 µL was prepared, consisting of 240 µL of phosphate buffer (0.1 M, pH 7.4), 10 µL of DTNB (0.01 M), 40 µL of tissue supernatant (the enzyme source), and 10 µL of ATCI (0.075 M). The absorbance was measured at 412nm using a UV-Vis spectrophotometer (Labman) over 5 minutes to assess the reaction kinetics. The enzyme activity was calculated using the following formula.
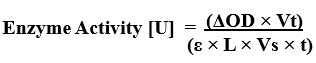
Enzyme Activity
AOD = change in absorbance, Vt = Total volume of reaction (in ml), E = Molar extinction coefficient, L = Path length (in cm), Vs = Sample volume used in assay (in ml), t = Time duration of reaction (in min)
Results
Table 1: Productivity Metrics (Mean ± SD) for Respiration, Gross, and Net Productivity in Three Urban Lakes
Sample Stations | Monsoon | Autumn | Winter | Spring | Summer |
Lingadheeranahalli Lake – Respiration Rate (mg/L) | |||||
Open | 0.47±0.81 | -3.3±5.59 | -0.45±0.92 | 2.2±1.13 | 1.57±2.04 |
Deposit | -0.60±3.14 | -7.0±9.05 | 0.55±0.07 | 2.05±0.78 | 0.80±0.20 |
Inlet | 0.30±0.52 | 7.7±10.29 | 1.2±1.41 | 2.3±0.85 | 3.10±3.28 |
Lingadheeranahalli Lake – Net productivity (mg/L) | |||||
Open | 8.87±1.45 | 10.6±2.90 | 0.25±9.97 | 2.55±0.49 | 0.73±3.76 |
Deposit | 14.67±11.72 | 15.3±0.94 | -1±7.07 | 2.75±1.48 | 2.83±0.25 |
Inlet | 1.13±1.96 | 8.4±9.40 | 6.5±2.40 | 3.5±0.85 | 0.63±4.27 |
Lingadheeranahalli Lake – Gross Productivity (mg/L) | |||||
Open | 9.33±0.64 | 7.3±2.69 | 3.7±5.37 | 4.75±1.63 | 2.30±1.73 |
Deposit | 12.73±6.36 | 7.7±7.17 | 2.45±3.04 | 4.8±0.71 | 3.63±0.38 |
Inlet | 1.43±2.48 | 1.1±1.52 | 7.1±1.84 | 5.8±1.70 | 3.73±1.22 |
Ullal Lake - Respiration Rate (mg/L) | |||||
Monsoon | Autumn | Winter | Spring | Summer | |
Open | 1.53±1.36 | -4.35±9.40 | 0.85±2.62 | 2.35±1.63 | 1.07±0.47 |
Deposit | -0.33±3.21 | -3.67±4.71 | -0.4±1.41 | 1.6±0 | 1.13±0.67 |
Inlet | 1.37±1.52 | -3.37±6.62 | 0.05±0.78 | 3.1±0.71 | 2.03±1.31 |
Ullal Lake – Net Productivity (mg/L) | |||||
Open | 8.67±3.06 | 11.5±6.36 | -0.2±3.39 | 3.6±1.13 | 0.90±0.75 |
Deposit | 14.20±8.45 | 10.6±5.09 | 1.35±2.47 | 4.15±1.06 | 1.10±1.31 |
Inlet | 20.90±13.65 | 14.55±8.98 | 4.05±3.75 | 2.15±1.63 | 1.97±0.74 |
Ullal Lake – Gross Productivity (mg/L) | |||||
Open | 10.20±1.71 | 2.15±10.11 | 3.7±1.70 | 5.95±0.49 | 1.97±0.31 |
Deposit | 13.87±6.82 | 13.93±0.09 | 3.55±0.21 | 5.75±1.06 | 2.10±1.15 |
Inlet | 22.27±12.13 | 19.23±4.29 | 5.7±2.26 | 5.25±2.33 | 4.00±0.69 |
Herohalli Lake – Respiration Rate (mg/L) | |||||
Open | 1.47±2.19 | -2.77±5.99 | 0.45±0.78 | 3.95±1.20 | 3.87±3.88 |
Deposit | 4.00±2.84 | -0.80±6.79 | 1.35±1.34 | 2.75±1.06 | 3.50±2.88 |
Inlet | 1.27±3.87 | 1.13±0.19 | 0.8±0.14 | 1.7±2.12 | 1.53±1.71 |
Herohalli Lake – Net Productivity (mg/L) | |||||
Open | 3.93±4 | 6.97±4.29 | 4.35±2.19 | -0.95±1.91 | -0.57±3.78 |
Deposit | 5.67±5.69 | 6.93±1.79 | 2.55±6.15 | 2.75±3.89 | -0.63±4.20 |
Inlet | 9.40±13.36 | 6.20±4.53 | 1.9±6.22 | 6.85±9.26 | 3.77±4.08 |
Herohalli Lake – Gross Productivity (mg/L) | |||||
Open | 5.40±6.12 | 1.20±5.94 | 7.2±0.42 | 3±0.71 | 3.30±0.10 |
Deposit | 9.67±8.52 | 3.53±8.67 | 6.85±0.64 | 5.5±2.83 | 2.83±1.27 |
Inlet | 10.67±9.50 | 7.33±4.71 | 5.65±1.91 | 8.55±7.14 | 5.30±3.24 |
Table 2: Chlorophyll Concentrations in Water Samples from Three Urban Lakes
Sample Stations | Monsoon | Autumn | Winter | Spring | Summer |
Lingadheeranahalli Lake - Chlorophyll A (mg/L) | |||||
Open | 8.63±0.55 | 9.7±0.07 | 9.9±1.77 | 7.75±0.07 | 6.41±3.72 |
Deposit | 12.20±0.46 | 10.2±0.5 | 9.4±0.3 | 8.5±0.1 | 13.3±5.8 |
Inlet | 8.07±0.6 | 9.7±0.1 | 8.9±0.4 | 7.8±0.1 | 17.9±12.7 |
Lingadheeranahalli Lake - Chlorophyll B (mg/L) | |||||
Open | 23.96±2.30 | 26.4±3.20 | 28.22±1.45 | 17.75±1.34 | 12.46±7.66 |
Deposit | 23.53±0.74 | 24.1±3.68 | 27.2±2.12 | 27.2±0.57 | 26.93±5.22 |
Inlet | 20.90±5.54 | 27.7±1.41 | 26.65±1.34 | 17.75±1.34 | 29.97±20.08 |
Lingadheeranahalli Lake - Total Chlorophyll (mg/L) | |||||
Open | 32.59±2.84 | 36.1±3.27 | 38.18±3.22 | 25.5±1.41 | 18.88±11.12 |
Deposit | 35.73±1.2 | 34.3±3.2 | 36.6±2.4 | 35.7±0.7 | 40.2±10.2 |
Inlet | 29±6.1 | 37.4±1.3 | 37.4±1.3 | 25.5±1.4 | 47.9±32.7 |
Ullal Lake - Chlorophyll A (mg/L) | |||||
Monsoon | Autumn | Winter | Spring | Summer | |
Open | 8.03± | 8.2±0.1 | 8.3±0.8 | 7.5±0.4 | 10.9±5.9 |
Deposit | 8.06± | 9.5±0.4 | 8.9±0.4 | 7.8±0.1 | 10.3±2.7 |
Inlet | 7.86± | 8.4±1.1 | 8.5±0.1 | 8.2±1.4 | 20.3±12.2 |
Ullal Lake - Chlorophyll B (mg/L) | |||||
Open | 23.33±2.22 | 27.7±1.41 | 18.1±5.66 | 17.75±1.34 | 10.90±16.19 |
Deposit | 20.73±5.25 | 22±7.35 | 27.2±2.12 | 27.2±0.57 | 18.92±4/42 |
Inlet | 23.37±5.77 | 21.4±8.20 | 17.75±1.34 | 26.2±0.71 | 38.92±15.39 |
Ullal Lake - Total Chlorophyll (mg/L) | |||||
Open | 31.4±2 | 35.9±1.3 | 26.4±4.8 | 25.3±0.9 | 21.8±21.8 |
Deposit | 28.8±5 | 31.5±7.8 | 36.1±2.5 | 35.0±0.5 | 29.2±7.1 |
Inlet | 31.2±6.1 | 29.8±9.3 | 26.3±1.2 | 34.4±0.7 | 58.6±26.3 |
Herohalli Lake - Chlorophyll A (mg/L) | |||||
Monsoon | Autumn | Winter | Spring | Summer | |
Open | 8.23± | 9.2±0.6 | 8.3±0.3 | 7.8±0.1 | 15.4±12.1 |
Deposit | 8.43± | 9.4±0.3 | 8.9±0.4 | 7.8±0.8 | 15.7±8.1 |
Inlet | 8.26± | 8.3±0.2 | 8.5±0.7 | 7.6±0.5 | 17.0±5.3 |
Herohalli Lake - Chlorophyll B (mg/L) | |||||
Open | 25.50±1.54 | 18.10±5.66 | 18.45±6.15 | 20.4±2.4 | 24.65±14.57 |
Deposit | 20.90±5.54 | 27.70±1.41 | 18.65±6.43 | 19.45±3.75 | 27.47±12.04 |
Inlet | 17.67±4.07 | 18.70±0 | 22.7±5.66 | 17.75±1.34 | 29.87±8.69 |
Herohalli Lake - Total Chlorophyll (mg/L) | |||||
Open | 32.7±1.5 | 27.3±6.2 | 26.8±6.4 | 28.2±2.3 | 40.1±26.7 |
Deposit | 29.3±5.2 | 37.1±1.1 | 27.6±6.9 | 27.3±4.6 | 43.1±19.9 |
Inlet | 25.9±4.3 | 27.0±0.2 | 31.0±6.4 | 25.3±0.8 | 46.9±13.1 |
The seasonal dynamics of respiration rate, gross productivity (PG), and net productivity (PN) across Herohalli, Ullal, and Lingadheeranahalli lakes exhibited significant spatiotemporal variation influenced by environmental and anthropogenic factors. Herohalli Lake exhibited the most consistent and elevated productivity, with peak net productivity at the inlet during the monsoon (9.40 ± 13.36 mg O2/L/day) and the highest gross productivity in spring (8.55 ± 7.14 mg O2/L/day). Negative PN values in spring and summer at the open station suggest increased respiration, potentially due to higher organic matter. Ullal Lake exhibited its highest productivity during the monsoon and autumn seasons, especially at the inlet, with gross productivity reaching 22.27 ± 12.13 mg O2/L/day and net productivity at 20.90 ± 13.65 mg O2/L/day. Conversely, productivity decreased in winter and summer, with net productivity falling to -0.2 ± 3.39 mg O2/L/day in winter. Lingadheeranahalli Lake exhibited lower and more erratic productivity, with modest gross productivity in spring (5.8 ± 1.70 mg O2/L/day) and net productivity fluctuating between -1.0 and 15.3 mg O2/L/day, indicating varying organic and nutrient loads. Chlorophyll concentrations supported these trends, with inlet stations during summer consistently showing the highest chlorophyll a and total chlorophyll levels across lakes. Ullal Lake recorded a peak total chlorophyll of 58.6 ± 26.3 µg/L at the inlet in summer, while Herohalli and Lingadheeranahalli followed, with inlet values of 46.9 ± 13.1 µg/L and 47.9 ± 32.7 µg/L, respectively. Chlorophyll-b concentrations were consistently higher than chlorophyll-a, with notable summer peaks at both inlet and deposit stations.
Overall, the monsoon and autumn seasons enhanced productivity across lakes. Conversely, despite elevated chlorophyll levels, summer showed lower or negative net productivity. Herohalli Lake ranked highest in overall productivity, followed by Ullal, while Lingadheeranahalli exhibited signs of ecological stress and eutrophic degradation.
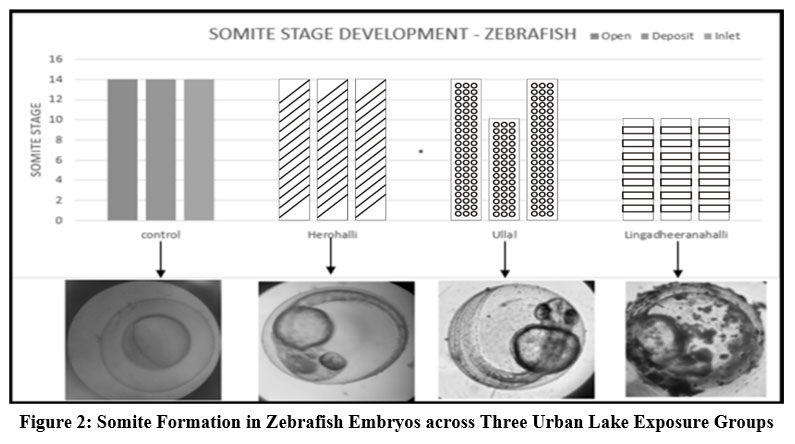 | Figure 2: Somite Formation in Zebrafish Embryos across Three Urban Lake Exposure Groups
|
Embryos exposed to Herohalli Lake water demonstrated normal somite development (Figure 2). They maintained a healthy heartbeat (Figure 3) of 113 to 108 beats per minute (bpm) during the torpedo stage, similar to the control group, indicating minimal toxicity. In contrast, Ullal Lake-exposed embryos exhibited mild developmental delays in somite segmentation and a reduced heart rate ranging from 89 to 127 bpm. Meanwhile, embryos exposed to Lingadheeranahalli Lake showed the most pronounced toxicological effects, with significant delays in somite formation, morphological abnormalities such as tail curvature, and a substantially lower heart rate, ranging from 21 to 99 bpm.
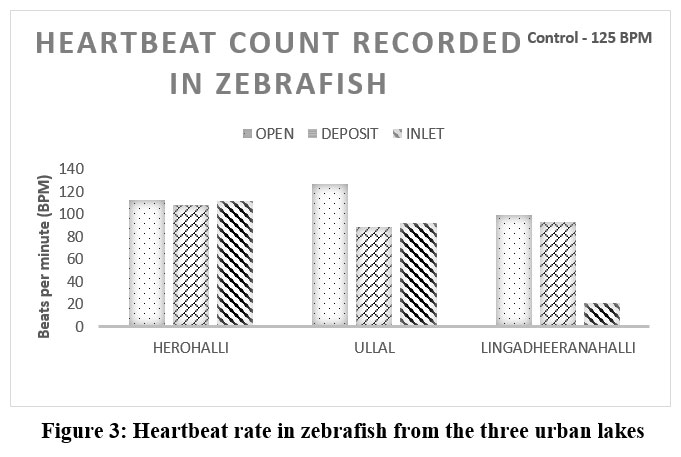 | Figure 3: Heartbeat rate in zebrafish from the three urban lakes
|
Table 3: Kruskal-Wallis and DSCF Statistical Results for Productivity and Chlorophyll Variables across Lakes, Stations, and Seasons
Kruskal-Wallis statistics indicating significance for respiration rate, gross productivity and net productivity between lakes through the light and dark bottle method, **p<0.05 | |||||||||
Lakes | Respiration Rate | Gross Productivity | Net Productivity | ||||||
X² | df | p | X² | df | p | X² | df | p | |
Herohalli Lake | 3.64 | 4 | 0.457 | 1.36 | 4 | 0.851 | 3.22 | 4 | 0.522 |
Ullal Lake | 5.38 | 4 | 0.250 | 9.83 | 4 | 0.043 | 8.72 | 4 | 0.069 |
Lingadheeranahalli Lake | 8.26 | 4 | 0.083 | 4.62 | 4 | 0.329 | 7.83 | 4 | 0.098 |
Kruskal-Wallis statistics indicating significance for chlorophyll a, chlorophyll b and total chlorophyll between lakes through the chlorophyll method, **p<0.05 | |||||||||
Lakes | Chlorophyll A | Chlorophyll B | Total Chlorophyll | ||||||
X² | df | p | X² | df | p | X² | df | p | |
Herohalli Lake | 8.79 | 4 | 0.067 | 2.97 | 4 | 0.562 | 3.06 | 4 | 0.547 |
Ullal Lake | 9.64 | 4 | 0.047 | 2.72 | 4 | 0.606 | 3.01 | 4 | 0.556 |
Lingadheeranahalli Lake | 9.39 | 4 | 0.052 | 5.88 | 4 | 0.208 | 4.41 | 4 | 0.353 |
Table 4: Kruskal-Wallis and DSCF Pairwise Comparisons between Lakes, Stations, and Seasons
Category / Comparison | Statistic / Pair | p |
C. Kruskal-Wallis Test | ||
Between Lakes | X² = 6.44 | 0.0399 |
Between Stations | X² = 3.10 | 0.2123 |
Between Seasons | X² = 14.21 | 0.0027 |
D. DSCF Pairwise Comparisons – Lakes | Herohalli vs Ullal | 0.3909 |
Herohalli vs Lingadheeranahalli | 0.0535 | |
Ullal vs Lingadheeranahalli | 0.0124 | |
E. DSCF Pairwise Comparisons – Seasons | Monsoon vs Summer | 0.0053 |
Autumn vs Summer | 0.0049 | |
Winter vs Summer | 0.0053 | |
Spring vs Summer | 0.0053 |
From table 3 and table 4, it is deduced that significant differences exist in total chlorophyll content between the lakes and also during various seasons, but no significant differences between the sampling stations, that is, open, deposit and inlet.
The acetylcholinesterase (AChE) activity measured in zebrafish exposed to lake water samples (Figure 4) varied significantly among the three lakes. The control group exhibited a baseline AChE activity of 0.0000345 µmol/min, serving as a reference for neurophysiological normalcy. Exposure to Herohalli Lake water markedly increased AChE activity to 0.0000822 µmol/min, indicating a possible stimulatory or compensatory neural response. Conversely, zebrafish exposed to Lingadheeranahalli Lake showed the lowest AChE activity of 0.0000263 µmol/min, suggesting neuro-inhibition. Fish exposed to Ullal Lake demonstrated AChE activity of 0.0000347 µmol/min, similar to the control, reflecting a relatively moderate impact on neurotoxicity.
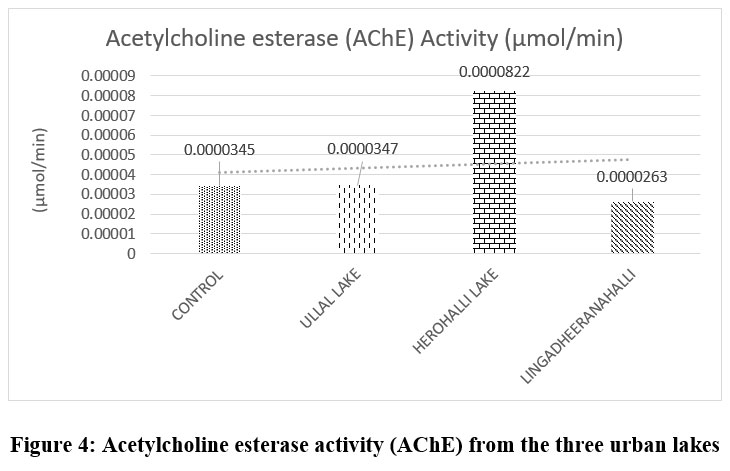 | Figure 4: Acetylcholine esterase activity (AChE) from the three urban lakes
|
Discussion
The present study employed a multimodal approach—combining seasonal limnological parameters with zebrafish bioassays—to assess ecological and neurotoxic stress in three urban lakes in North Bangalore. Our findings reveal a significant interplay between productivity parameters (gross and net productivity, chlorophyll levels) and biological indicators (heartbeat, somite formation, and AChE activity) across various seasonal cycles and lake environments. Primary Productivity and Chlorophyll Trends: Herohalli Lake consistently exhibited the highest productivity values, particularly during the monsoon and spring, as evidenced by peak gross productivity (10.67 ± 9.50 mg O2/L/day) and elevated chlorophyll concentrations at the inlet station (chlorophyll-a: 17.0 ± 5.3 µg/L; total chlorophyll: 46.9 ± 13.1 µg/L in summer). These findings align with Jones et al, who reported monsoonal peaks in nutrient influx and autotrophic activity in semi-urban water bodies.20 In contrast, Lingadheeranahalli Lake displayed highly variable and generally reduced productivity and chlorophyll levels, with gross productivity often falling below 6 mg O2/L/day and net productivity showing negative values during winter (-1.0 ± 7.07 mg O2/L/day). These trends suggest intense anthropogenic stress, nutrient overload, and ecological imbalance, reflecting the eutrophic degradation patterns.4,21 Reduced chlorophyll and productivity levels suggest ecological imbalance leading to algal blooms.22 The seasonal variations in productivity metrics highlight climate-induced changes such as shifts in rainfall, evaporation and stratification, which are increasingly affecting urban lakes.23,24
Zebrafish embryo toxicity test (ZEFT) remains economical with a high output compared to traditional toxicity tests.25 Zebrafish embryos exposed to Herohalli Lake exhibited standard developmental patterns and heartbeat values (113–108 bpm), corroborating the lake’s relatively healthier water quality, which aligns with ecological studies conducted on Herohalli Lake.26 In contrast, embryos exposed to Lingadheeranahalli showed developmental deformities, with heartbeats dropping as low as 21 bpm and delayed somite segmentation. These findings strongly indicate toxicity linked to pollutant accumulation and align with studies on Ibalur Lake.27
Acetylcholinesterase (AChE) activity is an active biomarker for the assessment of neurological toxicity to aquatic health.14,28 Zebrafish exposed to Herohalli water showed increased AChE activity (0.0000822 µmol/min), potentially reflecting cholinergic system compensation. In stark contrast, AChE activity was lowest in Lingadheeranahalli-exposed fish (0.0000263 µmol/min), revealing substantial neuro-inhibition, possibly due to pesticide or heavy metal contamination.11,13 Ullal Lake samples induced moderate physiological stress with AChE values near the control. These integrated findings underscore the importance of coupling physicochemical water quality monitoring with sensitive biological endpoints, such as zebrafish bioassays, for a more comprehensive understanding of aquatic health, particularly in urban ecosystems vulnerable to rapid anthropogenic encroachment.29,30
Conclusion
The integrated evaluation of three urban lakes through limnological assessments and zebrafish-based bioassays revealed insights into the seasonal productivity and ecological health of urban aquatic systems. Herohalli Lake exhibited good productivity and minimal biological toxicity, reflecting its relative ecological stability. Ullal Lake demonstrated moderate productivity and toxicity, suggesting transitional stress. However, Lingadheeranahalli Lake showed significant impairment, erratic productivity, low chlorophyll levels, and considerable neurotoxic effects on zebrafish embryos. The findings underscore the importance of combining traditional water quality assessments with biological markers such as AChE activity and developmental endpoints in zebrafish. This multimodal approach facilitates early detection of ecological degradation and aids in formulating targeted lake restoration and pollution management strategies.
Acknowledgement
The authors thank the School of Sciences, JAIN (Deemed to be University), for providing the laboratory infrastructure. We would also like to thank Dr. Jawahar Gandra and Dr. Monojit Bhatacharjee from the Department of Biotechnology and Genetics for their suggestions regarding the AChE assay.
Funding Sources
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Mathews P Raj: Conceptualization, Data collection, Methodology, Writing, and Statistical interpretation.
Reena Susan Philip Writing – Review & Editing.
References
- Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM. Global change and the ecology of cities. Science. 2008;319(5864):756-760. 2. doi:10.1126/science.1150195
CrossRef - Kumar A, Dua A. Water quality index for assessment of water quality of river Ravi at Madhopur (India). Glob J Environ Sci. 2010;8(1). doi:10.4314/gjes.v8i1.50824
CrossRef - Bai X, McPhearson T, Cleugh H, Nagendra H, Tong X, Zhu T. Linking urbanization and the environment: conceptual and empirical advances. Annu Rev Environ Resour. 2017;42(1):215-240. doi:10.1146/annurev-environ-102016-061128
CrossRef - Veenashree N, Nandini N, Kumar M. Nutrients load and eutrophication: An overview of Bengaluru urban lakes. Int J Res Advent Technol. 2018;6(11). http://www.ijrat.org/downloads/Vol-6/november-2018/paper%20ID-61129.pdf
- Timalsina R, Acharya S, ?urin, B, Awasthi MP, Pant R, Joshi G, Byanju R, Panthi K, Joshi S, Kumar T Tarun ,Saqr, A. An assessment of seasonal water quality in Phewa Lake, Nepal, by integrating geochemical indices and statistical techniques: A sustainable approach. Water. 2025;17(2):238. https://doi.org/10.3390/w17020238
CrossRef - Ramachandra TV, Sudarshan PB, Mahesh MK, Vinay S. Spatial patterns of heavy metal accumulation in sediments and macrophytes of Bellandur wetland, Bangalore. J Environ Manage. 2018;206:1204–1210. https://doi.org/10.1016/j.jenvman.2017.10.014
CrossRef - Bejagam V, Sharma A. Impact of climatic changes and anthropogenic activities on ecosystem net primary productivity in India during 2001–2019. Ecol Inform. 2022;70:101732. https://doi.org/10.1016/j.ecoinf.2022.101732
CrossRef - Jha S, Das J, Goyal MK. Assessment of risk and resilience of terrestrial ecosystem productivity under the influence of extreme climatic conditions over India. Sci Rep. 2019;9:18923. https://doi.org/10.1038/s41598-019-55067-0
CrossRef - Adhikary S, Chaturvedi S, Banerjee S, Basu S. Dependence of physicochemical features on marine chlorophyll analysis with learning techniques. In: Proceedings of International Conference on Sustainable Technologies for Computational Intelligence. Springer; 2021:361–373. https://doi.org/10.1007/978-3-030-79065-3_29
CrossRef - Durga Rao, G., Kanuri, V. V., Kumaraswami, M., Ezhilarasan, P., Rao, V. D., Patra, S. Ramu, K. Dissolved nutrient dynamics along the southwest coastal waters of India during northeast monsoon: A case study. Chem Ecol. 2017;33(1):1–18. https://doi.org/10.1080/02757540.2017.1287903
CrossRef - Truong L, Harper SL, Tanguay RL. Evaluation of embryo toxicity using the zebrafish model. In: Methods Mol Biol. 2011;691:271–279. https://doi.org/10.1007/978-1-60761-849-2_18
CrossRef - Engerer P, Plucinska G, Thong R, Trovò L, Paquet D, Godinho L. Imaging subcellular structures in the living zebrafish embryo. J Vis Exp. 2016;(110):e53456. https://doi.org/10.3791/53456
CrossRef - Rahman O, Jaman A, Shahjahan M, Jaman A. Impact of sex ratio on the spawning success of zebrafish in laboratory settings. Prog Agric. 2021;32:78–83. https://www.banglajol.info/index.php/PA/article/view/58481
CrossRef - Raj MP, Satishchandra N. Physico-chemical and microbiological characterization of recycled, open and eutrophicated lake water grabs and assessment of its effects on a vertebrate system Zebra fish (Danio rerio): A case study of Mallathahalli Lake, Bangalore, India. Biosci Biotechnol Res Asia. 2015;12(Special Edition 2). https://www.biotech-asia.org/vol12specialissue2/
CrossRef - Raj MP, Philip RS. Seasonal dynamics and ecological health of Ullal Lake in Bengaluru North, Karnataka, India, are introduced through a limnological perspective and physiological response assessment on zebrafish (Danio rerio). Asian J Biol Life Sci. 2024;13:297–307. https://www.ajbls.com/article/392
CrossRef - American Public Health Association. Standard Methods for the Examination of Water and Wastewater. 23rd ed. American Public Health Association; 2017. https://www.standardmethods.org/
- Nalini TJ, Suresh Kumar C, Geethanjali R, Prathibha KY, Priya M, Akshaya RS, Sonu D, Nandhini BS, Pragathi S. Quantitative extraction of chlorophyll a and chlorophyll b from eight medicinal plants using the Arnon method. Rev Electron Vet. 2024;25(1):3610–3616. DOI: 10.69980/redvet.v25i1.1650
CrossRef - Brand M, Granato M. Keeping and raising zebrafish. In: Schulte-Merker S, Nüsslein-Volhard C, editors. Zebrafish: A Practical Approach. Volume 261. Oxford: IRL Press; 1999. p. 7–37.
CrossRef - Ellman GL, Courtney KD, Andres V Jr, Featherstone RH. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
CrossRef - Jones J, McEachern P, Seo D. Empirical evidence of monsoon influences on Asian Lakes. Aquat Ecosyst Health Manag. 2009;12:129–137. https://doi.org/10.1080/14634980902907342
CrossRef - Raj MP, Philip RS. Unravelling the seasonal dynamics of limnological parameters and assessment of ecological health: A case study of Lingadheeranahalli Lake in Bangalore North, Karnataka, India. Biotechnol Res Asia. 2024;21(3). https://dx.doi.org/10.13005/bbra/3288
CrossRef - Wang H, Convertino M. Algal bloom ties: Systemic biogeochemical stress and Chlorophyll-a shift forecasting. Ecol Indic. 2023;154:110760. https://doi.org/10.1016/j.ecolind.2023.110760
CrossRef - Sterckx K, Delandmeter P, Lambrechts J, Jonathan Lambrechts, Eric Deleersnijder, Piet Verburg & Wim Thiery The impact of seasonal variability and climate change on Lake Tanganyika’s hydrodynamics. Environ Fluid Mech. 2023;23:103-123. https://doi.org/10.1007/s10652-022-09908-8
CrossRef - Sourav SK, Clements L, Nguyen TT, Buerkert A. Urbanization and seasonal effects on the lake ecosystem dynamics from 2002 to 2022 in Bengaluru, South India. Environ Challenges. 2024;15:100944. https://doi.org/10.1016/j.envc.2024.100944
CrossRef - OECD. Test No. 236: Fish Embryo Acute Toxicity (FET) Test. OECD Guidelines for the Testing of Chemicals. OECD Publishing; 2013. doi:10.1787/9789264203709-en
CrossRef - Raj MP, Philip RS. Seasonal dynamics of Herohalli Lake in North Bangalore, Karnataka, India: A limnological study of water. J Environ Biol. 2025;46:47–57. https://doi.org/10.22438/jeb/46/1/MRN-5300
CrossRef - Raj MP, Abraham AA, Reddy J. Effect of polluted water on Danio rerio (zebrafish) as a vertebrate model: A case study of Ibalur Lake, Bangalore, India. South Asian J Exp Biol. 2015;5:48–54. http://sajeb.org/vol-5-no-2-2015/
CrossRef - Lionetto MG, Caricato R, Calisi A, Giordano ME, Schettino T. Acetylcholinesterase as a biomarker in environmental and occupational medicine: new insights and future perspectives. Biomed Res Int. 2013;2013:321213. https://doi.org/10.1155/2013/321213
CrossRef - Li C, Chen Q, Zhang X, Snyder SA, Gong Z, Lam SH. An integrated approach with the zebrafish model for biomonitoring of municipal wastewater effluent and receiving waters. Water Res. 2018;131:33-44. https://doi.org/10.1016/j.watres.2017.12.017
CrossRef - Fender CL, Good SP, Garcia-Jaramillo M. An integrated approach to evaluating water contaminants and evaporation in agricultural water distribution systems. Ecotoxicol Environ Saf. 2024;287:117277. https://doi.org/10.1016/j.ecoenv.2024.117277
CrossRef






