Chitosan-Based Hydrogels for Sustainable Wastewater Treatment: A Comprehensive Review on Dye and Heavy Metal Removal
1
Centre of Excellence for Energy and Environmental Studies,
Deenbandhu Chhotu Ram University of Science and Technology,
Murthal, Sonepat,
Haryana
India
2
Department of Biomedical Engineering,
Deenbandhu Chhotu Ram University of Science and Technology,
Murthal, Sonepat,
Haryana
India
Corresponding author Email: jyoti.energy@dcrustm.org
DOI: http://dx.doi.org/10.12944/CWE.20.3.3
Copy the following to cite this article:
Sangwan A, Singh S, Rani J. Chitosan-Based Hydrogels for Sustainable Wastewater Treatment: A Comprehensive Review on Dye and Heavy Metal Removal. Curr World Environ 2025;20(3). DOI:http://dx.doi.org/10.12944/CWE.20.3.3
Copy the following to cite this URL:
Sangwan A, Singh S, Rani J. Chitosan-Based Hydrogels for Sustainable Wastewater Treatment: A Comprehensive Review on Dye and Heavy Metal Removal. Curr World Environ 2025;20(3).
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2025-07-25 |
|---|---|
| Accepted: | 2025-11-07 |
| Reviewed by: | 
 Balarak Davoud
Balarak Davoud
|
| Second Review by: |

 Shaimaa Fatih Ali
Shaimaa Fatih Ali
|
| Final Approval by: | Dr. Gangadhar Andaluri |
Introduction
Water is essential for all living organisms, yet, although it covers 71% of Earth's surface, only 3% is freshwater, with just 1% readily usable.1 Mountains, icebergs, glaciers, and polar snow represent potential locked-in reservoirs of remaining freshwater. Most freshwater is unavailable, and population and industrial expansion exacerbate this scarcity. The type of water pollution resulting from industrial activities depends on the facility's management. Water pollution is primarily due to the release of organic and inorganic pollutants into the aquatic environment. Wastewater from industries such as chemical manufacturing, battery production, metallurgy, and mining releases various coloring compounds (dyes), suspended particles, pesticides, and heavy metal ions.2 These contaminants pose a significant risk to the ecosystem. Approximately 10,000 tons of commercial dyes are consumed globally per annum.3 The textile sector accounts for 80% of global dye consumption.4 Dyes are also widely used in printing, food production, paper manufacturing, leather treatment, and pharmaceuticals. Their high-water solubility, chemical stability, and resistance to biodegradation allow them to persist in the environment for years. Many dyes are discharged into rivers and lakes without treatment, resulting in 280,000 tons of dye-contaminated wastewater emitted annually.5 Heavy metals such as Cu2+, Pb2+, Cd2+, Co2+, Mn2+, Ni2+, Zn2+, and Cr6+ are non-biodegradable, persistent, and pose a serious threat to human health and the environment. Various conventional methods exist, such as chemical coagulation, reverse osmosis, ion exchange (Figure 1), chlorination, and UV treatment, but have drawbacks such as high operating costs, secondary environmental issues, and complexity.6
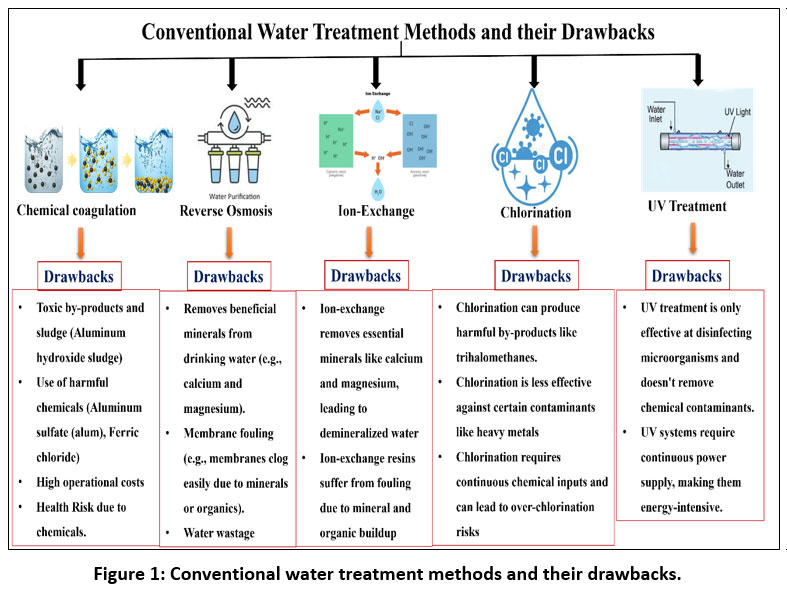 | Figure 1: Conventional water treatment methods and their drawbacks.
|
Given the rising threats of dye and heavy metal contamination in water sources, developing effective wastewater treatment systems is critical. Adsorption is the most practical and cost-effective method, offering superior efficacy among existing technologies. Common adsorbent agents, including zeolite, clay, alumina, and activated carbon, can remove various dangerous chemicals.7 Hydrogels are effective adsorbents because they can retain large quantities of water within their three-dimensional network, exhibit reversible expansion and contraction, and respond to various stimuli, such as temperature, electric current, and magnetic fields.8 Hydrogels are ideal for water treatment due to their biodegradability, ease of production using biobased and synthetic polymers, resistance to rapid thermal change, porous structure, large surface area, high swelling capacity, etc.9 Their hydrophilic groups also enable them to hold up to 10-20 times their mass in water, although they do not dissolve because of crosslinking.10 Hydrogel may be synthesized from either synthetic or natural polysaccharides. Generally, natural polymers such as starch, mucilage, chitosan, pectin, and so forth, are considered preferable because they are biodegradable, abundant, and easy to chemically modify.11 Their hydrophilic groups are capable of removing (heavy metals & dyes) via physical and chemical adsorption. Hydrogel matrices contain additives such as minerals, organic & inorganic molecules, metallic oxides, ions, and carbon-based additives, offering improved physicochemical properties, porous polymeric structure, recyclability, and multifunctionality.12
Unlike previous reviews that offer a generalized overview of chitosan hydrogels' performance, this review emphasizes a comprehensive quantitative comparison of adsorption capacities of removal efficiencies, and kinetic-isotherm behaviors across a wide range of pollutant types. By critically evaluating 31 articles from top-tier peer-reviewed publications, this discussion aims to identify the effects of molecular modifications, reinforcing agents, and processing conditions on adsorption performance. The novelty of this review lies in bridging experimental findings with practical applicability, offering an integrated perspective on performance optimization and real-world deployment potential. Moreover, this strategy not only enables a deeper understanding of chitosan hydrogel systems but also identifies areas that require further research to develop future generations of innovative clean water technologies.
Methodology
In this systematic review, it is imperative to highlight that its methodology is of tremendous significance, as it is intended to provide transparency, replicability, and integrity in the selection of studies, screening, and data extraction. A predetermined procedure for a systematic review is developed to align with its objectives, specifically those related to chitosan hydrogels in wastewater treatment. In this systematic review's methodology, inclusion and exclusion criteria, as well as predetermined data-extraction terms, have been clearly outlined.
Search strategy for data acquisition
Articles were searched across ScienceDirect, SpringerLink, Scopus, Wiley Online Library, and PubMed. This was done by searching for peer-reviewed publications of research studies published between 2019 and 2025. A selection of meticulously designed search terms was used to identify articles containing terms such as “Chitosan Hydrogels,” “Adsorption,” “Heavy Metals,” “Dye Removal,” and “wastewater treatment.” Boolean operators such as (AND/ OR) were utilized to ensure that all sources of articles were searched.
Inclusion and exclusion criteria
A paper qualified for the inclusion in this systematic review if it (i) focused on the synthesis, modification, or application of chitosan-based hydrogel for the adsorption of metal ions and dyes, (ii) provided quantitative data on adsorption capacities or efficiencies, and (iii) employed valid kinetic and isotherm models such as pseudo-first-order kinetic model or pseudo-second-order kinetic model, Langmuir model, or Freundlich model. Studies were excluded if they lacked sufficient quantitative information, were non-peer-reviewed, or focused on unrelated hydrogel systems.
Data extraction and analysis
The data extraction process was conducted meticulously to ensure the findings were accurate and reliable. Points considered in recording data include the type of hydrogel, the method of preparation, the modification of functionality, the adsorption capacity, the removal efficiency, the regeneration ability, and the kinetic or isotherm models applied. Statistics were used to calculate numerical ranges, the average adsorption value, and the kinetic applicability to provide a reliable overall performance assessment.
Quality assessment
The quality of the individual studies included in this systematic review was determined through a fair and objective analysis based on clear experimental designs, data replication, and adequate reporting of detailed study findings. Ideally, inconsistencies across studies or ambiguities in findings were resolved through joint discussion and verification by a team of reviewers, in an effort to maintain objectivity and increase validity within this systematic assessment.
Results
In total, 31 peer-reviewed articles were found that used chitosan hydrogel as an adsorbent for the removal of dyes and heavy metals from contaminated water systems. These studies collectively underline the strong adsorption potential, high removal efficiencies, and versatility of chitosan hydrogels and their composites. The final selection of articles was influenced by predefined criteria that included experimental studies reporting clear adsorption parameters, kinetic and isotherm modeling, and regeneration performance. The data synthesis provides a comprehensive overview of progress in applying chitosan hydrogel systems for wastewater treatment across a range of pollutants, including synthetic dyes and heavy metals.
In the first stage of database search, a total of 122 records were found. Among them, 62 documents were obtained from Scopus and 60 from PubMed. After excluding 65 non-relevant or duplicate records, 57 articles were selected for screening. A total of 26 articles were then excluded for lacking post-quantum adsorption data, being book chapters, not performing kinetic or isotherm modeling, or involving non-related hydrogel systems. A total of 31 qualitative studies met the criteria and were analyzed in detail. The PRISMA graphical illustration is shown in Figure 2.
 | Figure 2: The PRISMA flow diagram of a systematic review.
|
Natural polysaccharides
Natural polysaccharides are polymeric carbohydrate molecules composed of repeated monosaccharide units connected by glycosidic linkages. These naturally occurring biopolymers are utilized due to their bioresorbable properties, hypoallergenicity, non-immunogenicity, chemical stability, and non-toxicity, making them suitable for various applications.13 They are abundantly available in natural resources and can be processed at a relatively low cost. They possess various functional groups, such as hydroxyl, amine, and sulfonate, which facilitate chemical or physical bonding. Biocompatible hydrogels are commonly derived from polysaccharides, including chitin, starch, cassia gum, mucilage, pectin, guar gum, chitosan, and cellulose. The following section will provide a detailed examination of chitosan polysaccharides.
Chitosan
The chitosan is a carbohydrate extracted from algae & fungi and consists of an N-acetyl-D-glucosamine monomer attached to each other through a (1–4) linkage14 as shown in Figure 3. The white, powdery, flaky, iridescent, and non-toxic linear cationic polysaccharide is chitosan.15 The primary process for producing chitosan involves the deacetylation of chitin using an alkaline chemical. However, it is also a naturally occurring carbohydrate found in green algae and fungi. After cellulose, chitin is the most prevalent biopolymer. It is an intractable, weakly biodegradable polymer. Nonetheless, chitin's derivative, called chitosan, is a well-known biopolymer because it is non-toxic, biocompatible, less expensive, and soluble in acidic solutions. Eukaryotes such as fungi, insects, and crabs produce chitin.16 Chitin generally makes up about 20% to 30% of the weight of crustacean shell waste. Nonetheless, specific lobster genera have the highest chitin proportion of any chitin-containing animal, with chitin contents ranging from 60% to 75% by weight. The main characteristics of chitosan are changed by its molecular weight and degree of acetylation. These significantly impact functional and practical properties, such as dissolvability, biodegradability, the ability to impart the material structure, and other bioactive properties. Functional groups such as hydroxyl groups, secondary hydroxyl groups (OH) and primary amines are present in the chitosan molecule. These functional groups can be chemically altered without altering the degree of polymerization. Also, they allow chitosan to form hydrogen bonds and electrostatic interactions with heavy metals and dyes. Chitosan offers the advantages of a high recovery rate and reasonable cost compared to well-known materials like activated carbon for adsorption.17 Besides its antioxidant, fungistatic, and antimicrobial activity, it can be used as a substrate to produce films. The properties render it applicable in agriculture, biomedicine, and the environment. It can be used in the agricultural industry as a seed treatment agent and biopesticide to help plants resist fungal diseases. It can be used to safeguard the wine in the course of production, and it can also be used as a fining agent.18
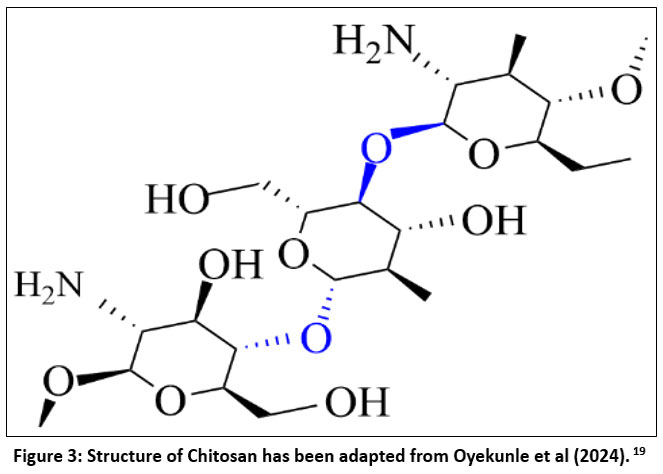 | Figure 3: Structure of Chitosan has been adapted from Oyekunle et al (2024).19
|
Other polysaccharides
Several polysaccharides, including cellulose, mucilage, starch, pectin, sodium alginate, glucan, guar gum, agar, and their derivatives, as well as chitosan, have enormous potential for adsorbing dyes and heavy metal ions, making them suitable for environmental remediation. Such polysaccharides are like mucilage in that they possess many natural origins, such as plants, algae, and microorganisms, which makes them sustainable and biodegradable.20 Starch is a polysaccharide made of glucose and is mainly obtained in fruits, pulses, roots, tubers (e.g., potatoes), and cereals. It consists of units of amylose and amylopectin.21 Cellulose is the most widespread polysaccharide, being derived from the extraction of cell walls of plants (e.g., cotton, wood), and containing B-1,4-linked glucose units. Pectin is a heteropolysaccharide of galacturonic acid produced by extracting the peel of citrus and apples.22 Sodium alginate is extracted using brown algae, i.e., Laminaria and Macrocystis, which contain mannuronic and guluronic acid units. B-glucan is derived from fungi, yeast, and cereals, i.e., oats and barley.23 Guar gum is a polysaccharide galactomannan of non-ionic nature extracted from the seeds of guar bean (Cyamopsis tetragonoloba).24 The agar is a polysaccharide comprising galactose and is obtained from red algae, such as Gelidium and Gracilaria. Enteromorpha polysaccharide is a sulfate polysaccharide that is derived from green algae Enteromorpha prolifera.25 The polysaccharides are divided into homopolysaccharides (e.g., cellulose, starch, glucan, agar) along with heteropolysaccharides (e.g., pectin, sodium alginate, guar gum, Enteromorpha polysaccharide). The functional groups give their adsorption properties as they contain hydroxyl (-OH), carboxyl (-COOH), and sulfate (-SO3H) groups (Table 1). The cellulose and starch contain hydroxyl groups that facilitate the hydrogen bonding with dyes, while pectin and sodium alginate contain carboxyl groups that enable the exchange of ions with heavy metal ions like Pb2+ and Cd2+.26 Sulfate groups in the Enteromorpha polysaccharide enhance the electrostatic interactions with cationic dyes, and the non-ionic features of guar gum enable the van der Waals interactions.27 The structural diversity and the large variety of functional groups in these polysaccharides make them useful platforms for hydrogel synthesis, which will boost the performance of mucilage-based systems in environmental applications. This is because of their broad accessibility from agricultural, algal, and microbial sources, which accentuates their efficacy in providing a renewable mode of removal.
Table 1: Polysaccharides, their parent groups, sources, and functional groups.
Polysaccharides | Parent group | Sources | Functional groups | Adsorption Mechanisms | References |
Enteromorpha polysaccharides | Heteropolysaccharides | Green algae (Enteromorpha prolifera) | Hydroxyl (-OH), sulfate (-SO3H) | Hydrogen bonding, electrostatic interaction | 28 |
Agar | Homopolysaccharides | Red algae (Gelidium, Gracilaria) | Hydroxyl (-OH), sulfate (-SO3H) | Hydrogen bonding, electrostatic interaction | 29 |
Guar Gum | Heteropolysaccharides | Guar bean seeds (Cyamopsis tetragonoloba) | Hydroxyl (-OH) | Hydrogen bonding, van der Waals | 24 |
Glucan (B- Glucan) | Homopolysaccharides | Fungi, yeast, cereals (oats, barley) | Hydroxyl (-OH) | Hydrogen bonding, van der Waals | 30 |
Sodium Alginate | Heteropolysaccharide | Brown algae (Laminaria, Macrocystis) | Carboxyl (-COOH), Hydroxyl (-OH) | Ion exchange, electrostatic interaction | 31 |
Pectin | Heteropolysaccharide | Fruit peels (Citrus, apple) | Carboxyl (-COOH), Hydroxyl (-OH) | Ion exchange, electrostatic interaction | 32 |
Starch | Homopolysaccharides | Fruits, pulses, roots/tubers (potatoes), cereals (corn, wheat) | Hydroxyl (-OH) | Hydrogen bonding, van der Waals | 33 |
Cellulose | Homopolysaccharides | Plant cell walls (cotton, wood) | Hydroxyl (-OH) | Hydrogen bonding, van der Waals | 34 |
Polysaccharide hydrogel for dye and heavy metal removal
The characteristics of biomaterial-based hydrogels mean they can be used in environmental regeneration programs. They could be beneficial in removing different pollutants from water bodies as they are able to absorb water. The most common type of biopolymer hydrogel is natural polysaccharides. Such materials are malleable and can undergo drastic changes to enhance their application.35 Consequently, they are the best at eliminating pollutants. Hydrogels based on polysaccharides have attracted the interest of many researchers. This section evaluates the use of chitosan-based hydrogels, reinforced with other materials as adsorbents for the removal of contaminants (heavy metals & dyes), along with their manufacturing processes and properties. The adsorption properties for dyes and heavy metals removal using natural polysaccharide-based hydrogel adsorption interactions that have already been examined will be discussed.
Removal of dyes using Chitosan hydrogel
Chitosan is a cationic macromolecule present in abundance in nature and can also be degraded by living things. It is often used to produce hydrogels.36 It has exceptional biological characteristics and degradability, which have led to its widespread application in tissue engineering, drug release, and skin restoration. It is also possible to consume in powdered or other edible forms, as -NH2 and OH groups are present in chitosan, this substance has a decent adsorption and chelation capacity toward metallic ions or anionic dyes.37 The pH level affects the amino group because it is protonated and deprotonated in basic and acidic environments. As per, the pendant group exhibits swelling behaviors when ionized, primarily when pH is below its pKa value. Chitosan's use in wastewater treatment is restricted by its low porosity, insolubility in water, lack of active sites, and low mechanical strength.38 Moreover, the mechanical durability of hydrogels derived from simple chitosan is low. To overcome constraints, chitosan-based hydrogels are synthesized using several organic and inorganic compounds, along with synthetic polymers, as additives. Wang et al., created a pH-CS interpenetrating hydrogel and CMC by cross-linking it with epichlorohydrin.39 They synthesized an amphoteric hydrogel, demonstrating swelling and adsorption in both acidic and basic environments. Several chitosan (CS) and carboxymethyl cellulose (CMC) solution ratios (8/2, 7/3, 6/4, and 5/5) were prepared using the solution casting process, mixed, and constantly swirled before the cross-linker was added. Investigations were conducted on removing Acid Orange II (ve) and MB (+ve). High swelling capacity carboxymethyl cellulose/chitosan (CMC5/CS5) hydrogel absorbed, 120 mg/g of MB, and 100 mg/g of Acid Orange II (AO II) at pH 11 & 2, respectively (Table 2). The adsorption process demonstrated that at an alkaline pH, CMC's COOH group ionizes to COO. & interacts in electrostatic interactions with MB. On the other hand, the chitosan NH2 NH3+ group demonstrated the most excellent affinity for AO II when protonated at pH levels above the acidic range. The hydrogel's potential uses across a broad pH spectrum are limited by its effectiveness, which is limited to specific pH ranges.39
Le et al. tested the removal of Congo red (CoR) and Brilliant Blue FCF (BBF) with a novel chitosan hydrogel activated with carbon dioxide. It was prepared using carbon dioxide gas passed through an adsorption tank containing a dye solution and chitosan. CO2-activated chitosan showed a greatly increased absorption capacity of the dye compared to chitosan in pure water.40 Both high temperatures and the initial adsorbate concentration had an impact on the dye adhesion on the hydrogel design. It was demonstrated that BBF bonded more rapidly than Congo Red (CoR) because the chitosan and -NH2 groups in CoR are electrostatically repelled. At temperatures ranging from 25 to 35 degrees Celsius, this hydrogel dissolves in an aqueous solution when the pH is lowered. Remarkably, when heated to 45 and 55 degrees Celsius, the hydrogel remained inflated and did not disintegrate in the solution. The hydrogel is stable at high pH values but unstable at low ones, and chitosan crosslinking is likely responsible for its increased stability between 45°C and 55°C.40 Hydrogel networks utilize clays to leverage their flexibility and wide adsorption capacity. Ferfera-Harrar et al., utilized Triton X-100 as a surfactant to develop a hydrogel nanocomposite (clay-based) for the removal of Basic Red 46.41 The matrix was hydrolyzed polyacrylamide, the polymer was chitosan, and the nanofiller was montmorillonite. By performing radical graft copolymerization of PAAm monomers, its stability and physicochemical properties improved, making it pH and temperature-sensitive. In a NaOH solution, hydrolysis of the hydrogel occurred, and it produced active groups, including amide anions, carboxylates (COO–), amines (NH2), and hydroxyl groups (OH) in the hydrogel. This procedure is convenient for the preparing of an ampholytic adsorption-like matrix. The nanocomposite's porosity and swelling were enhanced by adding a small amount of Montmorillonite (MMT) into the grafted matrix while applying a pore-generating agent, Triton X-100. With a 2-weight percent MMT addition, the hydrogel's adsorption ability rose from 1813 mg/g to 1550 mg/g. Physisorption was demonstrated by the isotherm data's perfect agreement with the Langmuir model (LM). The QM of the hydrogel has increased to 2 weight percent with the addition of MMT clay. Excess clay content lowered adsorption parameters (qe and R%). MMT particles occupy more network pores and interstitial spaces, reducing available space. MMT concentration increased network crosslinking due to hydrogen bonding interactions between clay hydroxyl groups and matrix components, such as chitosan (CTS) and hydrolyzed polyacrylamide (PAAm). Reduced network flexibility reduced water and dye penetration.41 Sharma et al., developed a superabsorbent composite hydrogel film consisting of citric acid-crosslinked Beta-cyclodextrin, chitosan, and polyvinyl alcohol (B-CD/Ch/PVA) that can effectively remove the dyes tartrazine (TG) and CoR from aqueous solutions. Fourier-transform infrared spectroscopic analysis verified that the functional groups -OH & -NH2 were present, while Field emission scanning electron microscopy demonstrated a rough, porous surface morphology.42 BET analysis confirmed the mesoporous characteristics of the hydrogel, revealing a mean pore diameter of 4.12 nm along with a surface area (SA) of 17.34 m²/g. The adsorption study was carried out using batch experiments & the Box-Behnken design model was used to optimize it. At a pH of neutral to slightly acidic, the hydrogel showed 95.38% and 96.3% removal of CoR & TG dye under ideal circumstances. The adsorption data supported monolayer adsorption, as evidenced by the matching of the Langmuir isotherm and the PSO kinetic model. The highest adsorption capabilities for CoR and TG have been identified as 1436.04 mg/g and 366.34 mg/g, respectively (Table 2). Endothermic or spontaneous adsorption is a product in a thermodynamic study, primarily driven by hydrogen bonds and electrostatic interactions between the dye molecules and the hydrogel matrix. B-CD/Ch/PVA hydrogel demonstrated significant reusability, retaining up to 85% of its removal efficiency across five adsorption–desorption cycles.42
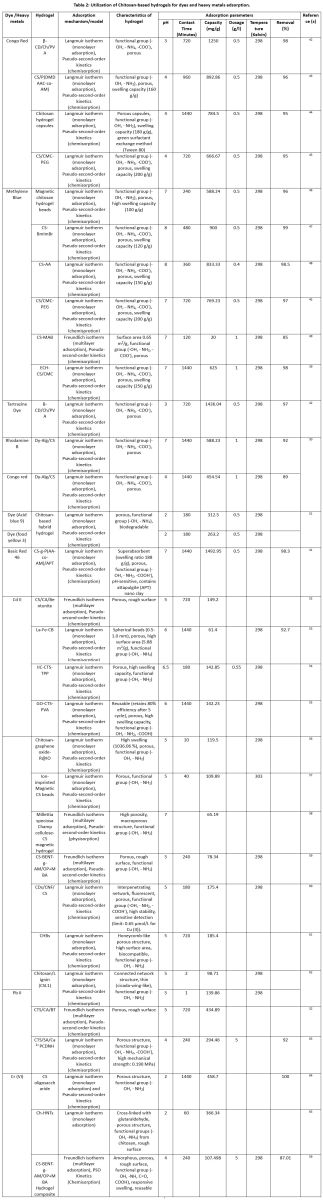 | Table 2: Utilization of Chitosan-based hydrogels for dyes and heavy metals adsorption.
|
Removal of heavy metals using Chitosan hydrogel
One hazardous heavy metal is lead, a risk to human health and aquatic ecosystems. Creating efficient, cost-effective, and sustainable materials for treating lead-contaminated wastewater is a significant challenge.66 To effectively remove hexavalent chromium (Cr6+) from aqueous solutions, Edebali et al.,65 produced chitosan-halloysite nanotube composite hydrogel beads (Ch-HNTs). Halloysite nanotubes (HNTs) were embedded in a chitosan matrix to create the hydrogel beads, and then glutaraldehyde was used to crosslink them for further stability. SEM and FTIR were confirmed the Ch-HNTs' structure along with functional characteristics. Batch adsorption experiments occurred out to evaluate the effectiveness of Cr6+ under various circumstances, like pH, contact duration, temperature, starting Cr6+ concentration, along with adsorbent dose. The findings showed that, with equilibrium duration of 60 minutes, optimum adsorption took place at pH 2. The experimental results showed the greatest has a 72.22 mg/g adsorption capacity, making it a good fit for the Langmuir isotherm model. For the removal of Cr6+ from watery settings, Ch-HNTs hydrogel beads show great promise as efficient adsorbents. Future studies will concentrate on how well they work in continuous-flow column systems and if they can be used to remove a wider variety of heavy metals (Table 2).65
Using a combination of blending, cross-linking, and grafting processes, Pavithra et al.,59 successfully created a biodegradable chitosan-based hydrogel composite (CS-BENT–g–AM/OP + MBA) to effectively remove the hazardous heavy metal ions Cr(VI) & Cu(II) from synthetic wastewater. The composite, which was made via chemically cross-linking radical-activated chitosan in polyacrylamide (AM) or N,N'-Methylene bisacrylamide (MBA), included OP as a natural addition. X-ray diffraction (XRD), Scanning electron microscopy, Energy dispersive X-ray analysis (SEM-EDAX), or FTIR have been employed to explain the morphological & structural characteristics of the hydrogel composite, confirming successful fabrication and functionalization. Through batch studies, the hydrogel's adsorption ability was systematically assessed by adjusting key factors, including the adsorbent dosage, contact time, pH and initial metal ion concentration. Atomic absorption spectroscopy (AAS) was used to determine the concentration of a metal ion before and after adsorption. The maximum removal of Cr(VI) & Cu(II) was reported at a contact time of 30 minutes, using 4 g of adsorbent at an initial metal ion concentration of 100 mg/g at a pH level of 4 and 5, respectively. The kinetic studies were best described by the PSO model, with the rate-determining step being chemisorption. The adsorption data also go well with the illustration of multilayer adsorption on a heterogeneous surface by application of the Freundlich model of isotherm. The hydrogel composite demonstrated impressive adsorption efficiencies of 82.47% for Cu(II) and 80.43% for Cr(VI) under ideal conditions, making it a potential, economical, and environmentally friendly adsorbent for treating metal-contaminated wastewater, particularly in high-volume applications.59
To enhance the capacity of biomass-based adsorbents to remove heavy metals through adsorption, Zhao et al.,62 developed a new chitosan and lignin hydrogel (CSL) using a straightforward one-pot synthesis method that employs the Mannich process. The produced CSL hydrogels performed better than traditional chitosan/lignin adsorbents made by the dissolution-drying process, which often exhibit low adsorption efficiency. This was especially true for Pb(II) - Cu(II) ion-selective adsorption from aqueous solutions. Among all its hydrogels, the optimized CSL1 exhibited very rapid adsorption kinetics, achieving equilibrium for Pb(II) in just one minute and for Cu(II) in under two minutes. With a maximal absorption capacity (mg/g) for Pb(II) of 98.71 & for Cu(II) of 139.86 (Table 2), they significantly outperformed a large number of previously documented adsorbents based on lignin and chitosan. Using X-ray photoelectron spectroscopy (XPS) & FTIR, the adsorption process was carefully examined, indicating that the main interaction causing metal ion capture is chemical complexation. The CSL hydrogel is proposed in this work as a potentially excellent green adsorbent with a high potential of being used in practical wastewater treatment and in the progress of developing environmentally friendly remedial technology due to its simple preparation technique, high adsorption capacity, and short equilibrium time.62
Discussion
The findings in this review show that chitosan-based hydrogels, especially when modified with cellulose and other strengthening agents, have great potential for sustainable wastewater treatment. The studies reviewed indicate that the adsorption capacities range from 263.2 to 1492.95 mg/g for dye adsorption and from 61.4 to 458.7 mg/g for metal ion adsorption, often exceeding those of current adsorbents such as activated carbon. Under optimized conditions, removal efficiencies of 95-99% have been reported in many studies for both dyes and metal ion contaminants. The high applicability of the pseudo-second-order kinetic model and the Langmuir isotherm in most works suggests a homogeneous adsorption mechanism involving chemisorption that leads to monolayer coverage. Thermodynamic results support the stability of these systems, showing endothermic, spontaneous adsorption processes suitable for various environmental conditions. Composite hydrogels made with carboxymethyl cellulose (CMC), sodium alginate, lignin, graphene oxide, or halloysite nanotubes. They show clear improvements in porosity, swelling, and strength. These improvements help with regeneration, achieving over 80% efficiency after five cycles of adsorption and desorption. Despite several advantages in wastewater treatment, most studies have been limited to lab tests, and all have used materials made in the lab. They do not include real-world wastewater, which often contains complex mixtures of contaminants. Also, beyond developing these hydrogel materials, there has been no research into whether they are cost-effective or how durable they are when used on a large scale. Addressing these gaps is important to turn lab results into practical applications. Future work should include pilot-scale testing, assessing the full life cycle, and combining chitosan hydrogels with other treatment methods, such as advanced oxidation or membrane filtration. This approach would improve treatment efficiency, operational stability, and economic viability, firmly positioning chitosan hydrogels as a key component in the future of green water remediation technologies.
Conclusion
This review emphasizes the strong potential of chitosan-based hydrogels as effective, environmentally friendly materials for removing dyes and heavy metals from wastewater. They have high absorption capacities (263.2-1492.95 mg/g for dyes and 61.4-458.7 mg/g for metals) and removal efficiencies (over 95-99%), showing better performance than traditional materials. The prevalence of PSO kinetics and the Langmuir isotherm indicates a coherent chemisorption process. The incorporation of reinforcements, including CMC, sodium alginate, lignin, and graphene oxide, enhanced structural stability and reusability. Although laboratory results are promising, subsequent research must prioritize applying these materials to actual wastewater, assessing their long-term performance, and analyzing their cost-effectiveness to facilitate the transition from research to large-scale implementation.
Acknowledgment
The author gratefully acknowledges the Centre of Excellence for Energy and Environment Studies at Deenbandhu Chhotu Ram University of Science and Technology, Murthal, for providing the necessary facilities and academic environment that enabled the completion of this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions:
Anju Sangwan has been written original draft,
Jyoti Rani and Sarita Singh Supervised, reviewed and edited the manuscript.
References
- Mishra RK. Fresh water availability and its global challenge. Br J Multidiscip Adv Stud. 2023;4(3):1-78.
CrossRef - Oladimeji TE, Oyedemi M, Emetere ME, Agboola O, Adeoye JB, Odunlami OA. Review on the impact of heavy metals from industrial wastewater effluent and removal technologies. Heliyon. 2024;10(23).
CrossRef - Hagan E, Poulin J. Statistics of the early synthetic dye industry. Herit Sci. 2021;9(1).
CrossRef - Farhana K, Kadirgama K, Mahamude ASF, Mica MT. Energy consumption, environmental impact, and implementation of renewable energy resources in global textile industries: an overview towards circularity and sustainability. Mater Circ Econ. 2022;4(1).
CrossRef - Domingues NS, Romão ÉL, Alvim DS, Marques JP, Rodrigues VGS, Kasemodel MC. Use of Construction and Demolition Waste for the Treatment of Dye-Contaminated Water Toward Circular economy. Water Air Soil Pollut. 2024;235(10):663.
CrossRef - Levy-Ontman O, Yanay C, Alfi Y, Paz-Tal O, Wolfson A. Selective sorption of heavy metals by renewable polysaccharides. Polymers. 2023;15(22):4457.
CrossRef - Velarde L, Nabavi MS, Escalera E, Antti ML, Akhtar F. Adsorption of heavy metals on natural zeolites: A review. Chemosphere. 2023;328:138508.
CrossRef - Guo Y, Bae J, Fang Z, Li P, Zhao F, Yu G. Hydrogels and Hydrogel-Derived Materials for Energy and Water Sustainability. Chem Rev. 2020;120(15):7642-7707.
CrossRef - Van Tran V, Park D, Lee YC. Hydrogel applications for adsorption of contaminants in water and wastewater treatment. Environ Sci Pollut Res. 2018;25(25):24569-24599.
CrossRef - Radoor S, Karayil J, Jayakumar A, Kandel D.R, Kim J.T, Siengchin S, Lee J. Recent advances in cellulose-and alginate-based hydrogels for water and wastewater treatment: A review. Carbohydr Polym. 2024;323:121339.
CrossRef - Berradi A, Aziz F, Achaby ME, Ouazzani N, Mandi L. A comprehensive review of polysaccharide-based hydrogels as promising biomaterials. Polymers. 2023;15(13):2908.
CrossRef - Wu Y, Parandoust A, Sheibani R, Kargaran F, Khorsandi Z, Liang Y, Xia C, Van Le Q. Advances in gum-based hydrogels and their environmental applications. Carbohydr Polym. 2023;318:121102.
CrossRef - Panda J, Mishra AK, Mohanta YK, Patowary K, Rauta PR, Mishra B. Exploring Biopolymer for Food and Pharmaceuticals Application in the Circular Bioeconomy: An Agro-Food Waste-to-Wealth Approach. Waste Biomass Valor. 2024;15(10):5607-5637.
CrossRef - Singh S. Research on polyethylene glycol, crosslinked polyethylene glycol & polyethylene glycol chitosan conjugate coating for biomedical application. IJRTE. 2019;8:2921-2925.
CrossRef - Sharma S, Bhende M. An overview: non-toxic and eco-friendly polysaccharides—its classification, properties, and diverse applications. Polym Bull. 2024;81(14):12383-12429.
CrossRef - Perelygin VV, Zharikov MV, Zmitrovich IV, Nekrasova TA. Chitin and Its Derivative Chitosan: Distribution in Nature, Applications, and Technology Research (A Review). Int J Med Mushrooms. 2024;26.
CrossRef - Riseh RS, Vazvani MG, Kennedy JF. The application of chitosan as a carrier for fertilizer: A review. Int J Biol Macromol. 2023;252:126483.
CrossRef - Hafeez S, Islam A, Durrani AK, Butt MT, Rehmat S, Khurshid A, Khan SM. Fabrication of pectin-based stimuli responsive hydrogel for the controlled release of ceftriaxone. Chem Pap. 2023;77(4):1809-1819.
CrossRef - Oyekunle DT, Nia MH, Wilson LD. Recent Progress on the Application of Chitosan, Starch and Chitosan–Starch Composites for Meat Preservation—A Mini Review. J Compos Sci. 2024;8(8):302.
CrossRef - Shiam MdAH, Islam MS, Ahmad I, Haque SS. A review of plant-derived gums and mucilages: Structural chemistry, film forming properties and application. J Plast Film Sheeting. 2025;41(2):195-237.
CrossRef - Nath R, Dutta K. Sources of starch and its morphology: A review. Interdiscip Res Technol Manag. 2024:402-411.
CrossRef - De Souza TS, Kawaguti HY. Cellulases, hemicellulases, and pectinases: Applications in the food and beverage industry. Food and Bioprocess Technol. 2021;14(8):1446-1477.
CrossRef - Singla A, Gupta OP, Sagwal V, Kumar A, Patwa N, Mohan N, Ankush, Kumar D, Vir O, Singh J, Kumar L. Beta-glucan as a soluble dietary fiber source: Origins, biosynthesis, extraction, purification, structural characteristics, bioavailability, biofunctional attributes, industrial utilization, and global trade. Nutrients. 2024;16(6):900.
CrossRef - Tahmouzi S, Meftahizadeh H, Eyshi S, Mahmoudzadeh A, Alizadeh B, Mollakhalili-Meybodi N, Hatami M. Application of guar (Cyamopsis tetragonoloba L.) gum in food technologies: A review of properties and mechanisms of action. Food Sci Nutr. 2023;11(9):4869-4897.
CrossRef - Yu Y, Li Y, Du C, Mou H, Wang P. Compositional and structural characteristics of sulfated polysaccharide from Enteromorpha prolifera. Carbohydr Polym. 2017;165:221-228.
CrossRef - Sharma A, Sharma PK, Malviya R. Utilization of Polysaccharides and their Derivatives in the Removal of Metal Ions: Role and Recent Advancement. CMS. 2021;13(2):77-96.
CrossRef - Ma X, Duan D, Chen J, Xie B. Structure and Adsorption Performance of Cationic Entermorpha prolifera Polysaccharide-Based Hydrogel for Typical Pollutants: Methylene Blue, Cefuroxime, and Cr (VI). Gels. 2022;8(9):546.
CrossRef - Zhang H, Shi LWE, Zhou J. Recent developments of polysaccharide-based double-network hydrogels. J Polym Sci. 2023;61(1):7-43.
CrossRef - Agars: Properties and Applications. In: Polysaccharides. 1st ed. Wiley; 2021:75-93.
CrossRef - Izydorczyk MS. Functional properties of cereal cell wall polysaccharides. In: Carbohydrates in Food. CRC Press; 2017:215-278.
- Abka-Khajouei R, Tounsi L, Shahabi N, Patel AK, Abdelkafi S, Michaud P. Structures, properties and applications of alginates. Mar Drugs. 2022;20(6):364.
CrossRef - Das SK. Orange peel derived natural and modified pectin for the removal of heavy metals from aqueous solution. 2019.
- Nath R, Dutta K. Sources of starch and its morphology: A review. Interdiscip Res Technol Manag. 2024:402-411.
CrossRef - Jarvis MC. Hydrogen bonding and other non-covalent interactions at the surfaces of cellulose microfibrils. Cellulose. 2023;30(2):667-687.
CrossRef - Zhao C, Liu G, Tan Q, Gao M, Chen G, Huang X, Xu X, Li L, Wang J, Zhang Y, Xu D. Polysaccharide-based biopolymer hydrogels for heavy metal detection and adsorption. J Adv Res. 2023;44:53-70.
CrossRef - Mishra A, Omoyeni T, Singh PK, Anandakumar S, Tiwari A. Trends in sustainable chitosan-based hydrogel technology for circular biomedical engineering: a review. Int J Biol Macromol. 2024:133823.
CrossRef - Echazú MIA, Olivetti CE, Anesini C, Perez CJ, Alvarez GS, Desimone MF. Development and evaluation of thymol-chitosan hydrogels with antimicrobial-antioxidant activity for oral local delivery. Mater Sci Eng C. 2017;81:588-596.
CrossRef - Bhatt P, Joshi S, Bayram GMU, Khati P, Simsek H. Developments and application of chitosan-based adsorbents for wastewater treatments. Environ Res. 2023;226:115530.
CrossRef - Wang W, Hu J, Zhang R, Yan C, Cui L, Zhu J. A pH-responsive carboxymethyl cellulose/chitosan hydrogel for adsorption and desorption of anionic and cationic dyes. Cellulose. 2021;28(2):897-909.
CrossRef - Le HQ, Sekiguchi Y, Ardiyanta D, Shimoyama Y. CO2 -Activated Adsorption: A New Approach to Dye Removal by Chitosan Hydrogel. ACS Omega. 2018;3(10):14103-14110.
CrossRef - Ferfera-Harrar H, Benhalima T, Sadi A. Development of functional chitosan-based superabsorbent hydrogel nanocomposites for adsorptive removal of Basic Red 46 textile dye. Polym Bull. 2022;79(8):6141-6172.
CrossRef - Sharma P, Sharma M, Yadav L, Agarwal M, Gupta R. Sustainable solution for wastewater management: Fabrication of cost-effective ?-cyclodextrin incorporated chitosan polyvinyl alcohol composite hydrogel film for the efficient adsorption of anionic congo red and tartrazine dyes. Sep Purif Technol. 2025;355:129750.
CrossRef - Liu H, Zhang H, Ye Z, Xiong G. Efficient removal of anionic dye congo red by Chitosan/Poly (dimethyl diallyl ammonium chloride-co-acrylamide) composite hydrogel. Int J Biol Macromol. 2025;294:139462.
CrossRef - Chatterjee S, Tran HN, Godfred OB, Woo SH. Supersorption Capacity of Anionic Dye by Newer Chitosan Hydrogel Capsules via Green Surfactant Exchange Method. ACS Sustainable Chem Eng. 2018;6(3):3604-3614.
CrossRef - Zhu H, Chen S, Duan H, He J, Luo Y. Removal of anionic and cationic dyes using porous chitosan/carboxymethyl cellulose-PEG hydrogels: Optimization, adsorption kinetics, isotherm and thermodynamics studies. Int J Biol Macromol. 2023;231:123213.
CrossRef - Jamali M, Akbari A. Facile fabrication of magnetic chitosan hydrogel beads and modified by interfacial polymerization method and study of adsorption of cationic/anionic dyes from aqueous solution. J Environ Chem Eng. 2021;9(3):105175.
CrossRef - Karimi-Maleh H, Ranjbari S, Tanhaei B, Ayati A, Orooji Y, Alizadeh M, Karimi F, Salmanpour S, Rouhi J, Sillanpää M, Sen F. Novel 1-butyl-3-methylimidazolium bromide impregnated chitosan hydrogel beads nanostructure as an efficient nanobio-adsorbent for cationic dye removal: Kinetic study. Environ Res. 2021;195:110809.
CrossRef - Mustafa FH, EIRab EKMG, Kamel RM, Elshaarawy RFM. Cost-effective removal of toxic methylene blue dye from textile effluents by new integrated crosslinked chitosan/aspartic acid hydrogels. Int J Biol Macromol. 2023;248:125986.
CrossRef - Demirezen DA, Y lmaz DD, Y ld z Y. Magnetic chitosan/calcium alginate double-network hydrogel beads: Preparation, adsorption of anionic and cationic surfactants, and reuse in the removal of methylene blue. Int J Biol Macromol. 2023;239:124311.
CrossRef - Malik SA, Dar AA, Banday JA. Rheological, morphological and swelling properties of dysprosium-based composite hydrogel beads of alginate and chitosan: A promising material for the effective cationic and anionic dye removal. Colloids Surf Physicochem Eng Asp. 2023;663:131046.
CrossRef - Pauletto PS, Gonçalves JO, Pinto LAA, Dotto GL, Salau NPG. Single and competitive dye adsorption onto chitosan–based hybrid hydrogels using artificial neural network modeling. J Colloid Interface Sci. 2020;560:722-729.
CrossRef - Lin Z, Yang Y, Liang Z, Zeng L, Zhang A. Preparation of chitosan/calcium alginate/bentonite composite hydrogel and its heavy metal ions adsorption properties. Polymers. 2021;13(11):1891.
CrossRef - Lan Z, Lin Y, Yang C. Lanthanum-iron incorporated chitosan beads for adsorption of phosphate and cadmium from aqueous solutions. Chem Eng J. 2022;448:137519.
CrossRef - Babakhani A, Sartaj M. Synthesis, characterization, and performance evaluation of ion-imprinted crosslinked chitosan (with sodium tripolyphosphate) for cadmium biosorption. J Environ Chem Eng. 2022;10(2):107147.
CrossRef - Li C, Yan Y, Zhang Q, Zhang Z, Huang L, Zhang J, Xiong Y, Tan S. Asorption of Cd2+ and Ni2+ from Aqueous Single-Metal Solutions on Graphene Oxide-Chitosan-Poly(vinyl alcohol) Hydrogels. Langmuir. 2019;35(13):4481-4490.
CrossRef - Ali A, Khan S, Garg U, Luqman M, Bhagwath SS, Azim Y. Chitosan-based hydrogel system for efficient removal of Cu [II] and sustainable utilization of spent adsorbent as a catalyst for environmental applications. Int J Biol Macromol. 2023;247:125805.
CrossRef - Ma L, Zheng Q. Selective adsorption behavior of ion-imprinted magnetic chitosan beads for removal of Cu (II) ions from aqueous solution. Chin J Chem Eng. 2021;39:103-111.
CrossRef - Chen X, Huang Z, Luo SY, Zong MH, Lou WY. Multi-functional magnetic hydrogels based on Millettia speciosa Champ residue cellulose and Chitosan: Highly efficient and reusable adsorbent for Congo red and Cu2+ removal. Chem Eng J. 2021;423:130198.
CrossRef - Pavithra S, Thandapani G, Alkhamis HH, Alrefaei AF, Almutairi MH. Batch adsorption studies on surface tailored chitosan/orange peel hydrogel composite for the removal of Cr (VI) and Cu (II) ions from synthetic wastewater. Chemosphere. 2021;271:129415.
CrossRef - Chen X, Song Z, Yuan B, Li X, Li S, Nguyen T.T, Guo M, Guo Z. Fluorescent carbon dots crosslinked cellulose Nanofibril/Chitosan interpenetrating hydrogel system for sensitive detection and efficient adsorption of Cu (II) and Cr (VI). Chem Eng J. 2022;430:133154.
CrossRef - Yang J, Liu R, Sun Z, Zhang Y, Ju Y, Li X, Wang J, Gong Y. Synthesis of honeycomb-like citric acid-crosslinked chitosan hydrogel beads (cCHBs): Insight into structural characteristics of Cu (II)-loaded cCHBs (cCHBs-Cu (II)). Int J Biol Macromol. 2025:140244.
CrossRef - Zhao X, Zhao H, Mei A, Peng L, Sun J. Novel chitosan/lignin hydrogel prepared by the Mannich reaction for Pb (II) and Cu (II) removal from aqueous solution. Int J Biol Macromol. 2025;285:138177.
CrossRef - Tang S, Yang J, Lin L, Peng K, Chen Y, Jin S, Yao W. Construction of physically crosslinked chitosan/sodium alginate/calcium ion double-network hydrogel and its application to heavy metal ions removal. Chem Eng J. 2020;393:124728.
CrossRef - Mei J, Zhang H, Li Z, Ou H. A novel tetraethylenepentamine crosslinked chitosan oligosaccharide hydrogel for total adsorption of Cr (VI). Carbohydr polym. 2019;224:115154.
CrossRef - Edebali S, Kahraman HT, Karakaya MC. Removal of Hexavalent Chromium (Cr6+) from Aqueous Solutions by Chitosan-halloysite Nanotubes Composite Hydrogel Beads. Curr Anal Chem. 2025;21(2):165-173.
CrossRef - Chowdhury IR, Chowdhury S, Mazumder MAJ, Al-Ahmed A. Removal of lead ions (Pb2+) from water and wastewater: a review on the low-cost adsorbents. Appl Water Sci. 2022;12(8):185.
CrossRef






