Actinomycetes as a Promising Tool for Plastic and Hydrocarbon Biodegradation
1
Department of Microbiology,
Atmiya University,
Rajkot,
Gujarat
India
Corresponding author Email: mousumi.das@atmiyauni.ac.in
DOI: http://dx.doi.org/10.12944/CWE.20.2.17
Copy the following to cite this article:
Yagnik U. B, Das M. B. Actinomycetes as a Promising Tool for Plastic and Hydrocarbon Biodegradation. Curr World Environ 2025;20(2). DOI:http://dx.doi.org/10.12944/CWE.20.2.17
Copy the following to cite this URL:
Yagnik U. B, Das M. B. Actinomycetes as a Promising Tool for Plastic and Hydrocarbon Biodegradation. Curr World Environ 2025;20(2).
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2025-04-22 |
|---|---|
| Accepted: | 2025-06-25 |
| Reviewed by: | 
 Shaista Shafiq
Shaista Shafiq
|
| Second Review by: |

 Anil Deshpande
Anil Deshpande
|
| Final Approval by: | Dr. Hiren B Soni |
Introduction
In the fields of chemistry and material sciences, the synthesis of polymers like plastic from crude oil was revolutionary. This discovery opened a new doorway for the production of the most resistant and durable material, such as plastic.
Hydrocarbons are organic compounds made up solely of hydrogen and carbon atoms. In many industrial operations, they are vital raw materials and the main components of fossil fuels, such as coal, oil, and natural gas.
There have been several reports that confirm the microbial degradation of LDPE and hydrocarbons by several microbial species. The bacterial species reported for LDPE degradation include: Acinetobacter Brevibacillus, Rhodococcus, Micrococcus, and Pseudomonas.
Materials and Methods
Review literature
To conduct research and review literature, a comprehensive search was conducted across major scientific databases such as PubMed, ScienceDirect, SpringerLink, Google Scholar, and ResearchGate to find relevant literature. Several keywords were used: "LDPE biodegradation," "plastic degrading bacteria," "actinomycetes and polyethylene," "bioremediation of plastics," "plastic dumping site microbiota," "hydrocarbon degrading microorganisms," as well as "FTIR in polymer degradation." Following an initial screening of titles and abstracts, about 50 papers were chosen for full-text review. The most significant 36 studies were rigorously analysed and cited in the current study.
Isolation of actinomycetes
To isolate actinomycetes, soil samples were collected from numerous garbage disposal sites in Rajkot, India. Four distinct locations inside Rajkot City were selected for sample collection, which were Aji Dam, Vagodad, Navagam, and Sadhuvasvani Road. The sample collection was done by collecting soil from the three to five centimetres, and the samples were collected in a sterile zip bag. The samples of soil were dried at 70 °C for two hours for the isolation of the actinomycetes from the soil. In addition, 1 g of the heated samples was serially diluted and applied to sterile starch casein agar plates (10 g of soluble starch, 2 g K2HPO4, 2 g KNO3, 0.3 g casein, and 0.05 g MgSO4.7H2O, 0.01 g FeSo4.7H2O, 0.02 CaCO3 in 1 L of distilled water). The actinomycetes colonies were recognized by physical characteristics and Gram staining following seven days of incubation at 30 °C. They were then streaked further to preserve them on starch casein agar plates.
Screening for LDPE degradation
Screening of the isolated actinomycetes was carried out by spreading them on sterile minimal salt medium (MSM) containing NaNo3 (2 g), MgSo4 (0.5 g), Kcl (0.5 g), FeSo4 (0.01g), KH2Po4 (0.14 g), K2HPo4 (1.2 g), and Yeast extract (0.02 g) adding 1% LDPE powder as a carbon source. Followed by observing the growth of isolates in MSM broth with 1 % LDPE powered at 600nm till 7 days at 30 °C in shaking conditions at 120 rpm.
Screening for Alkane degradation
Screening of isolated actinomycetes for alkane degradation was carried out by observing the growth of isolates in minimal salt broth, adding 1% n-hexadecane as a model alkane at 600nm for 7 days at 30 °C in shaking conditions at 120 rpm.
Lab Scale Degradation LDPE Sheets
The lab-scale degradation of LDPE Sheets regularly used for packaging purposes was carried out by cutting the LDPE sheets into 2 by 2 cm, followed by exposing the LDPE sheets to the potent isolates for 30 days in sterile MSM medium at 30 °C in shaking conditions at 120 rpm. Followed by the incubation period, LDPE sheets were washed with 2% SDS for 3-4 Hours, and afterwards it was cleaned up with deionized water and dried at 60 °C overnight. After the incubation, the dry weight of the sheets was measured by the following formula.25
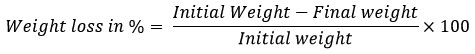
FTIR analysis of LDPE Sheets
The changes in the chemical structure of the LDPE sheets were observed by comparing the control and LDPE sheets inoculated with the potent isolates by FTIR spectroscopy at 500-4000 cm-1.
Results
Isolation of actinomycetes
Researchers have identified plastic dumping sites as important repositories of diverse microbiota, many of which can break down a variety of environmental contaminants, including plastics and hydrocarbons. In this work, 50 actinomycete strains were identified from soils at four separate dumping sites in Rajkot, India. Soil samples were collected and put on sterile Starch Casein Agar (SCA) plates. After a 7 to 14 day incubation period, separate colonies were discovered and isolated: 14 from Aji Dam, 15 from Vagodad, 11 from Navagam, and 10 from Sadhu Vasvani Road. The isolates were identified by microscopic examination as non-motile, filamentous, Gram-positive bacteria that belonged to the actinomycetes group. The colouration of the colonies ranged from grey (e.g., PUA 11, PUA 35), light greyish (e.g., PUA 9, PUA 15), to white (e.g., PUA 6, PUA 20). This variance in colony morphology and pigmentation gives preliminary information about the taxonomic richness of the isolated strains, which may correlate with differences in biodegradation capacity.
Screening of isolates for LDPE degradation
The primary screening for LDPE degradation was carried out by spreading the culture of isolates on MSM plates containing 1% LDPE as the only source of carbon. The findings of primary screening revealed that out of 50 different actinomycetes from the plastic dumping sites, only 29 isolates were able to grow well on MSM with LDPE powder. Whereas 12 isolates showed moderate to little growth in the presence of LDPE powder, 8 isolates were unable to use LDPE powder as a source of carbon; hence, in MSM plates, these isolates showed no signs of growth. Secondary screening for LDPE degradation was carried out by observing the growth of isolates in MSM broth with 1% LDPE powder at 600nm up to 7 days. Out of 29 isolates, only 10 actinomycetes showed a potential growth in the presence of LDPE, which is represented in Figure 1. The maximum growth O.D observed initially was by PUA 11 (0.071 + 0.004) and PUA 20 (0.071+ 0.01). At the end of the incubation the maximum growth was observed in PUA 35 (0.378 + 0.006) followed by PUA 20 (0.370 + 0.005), PUA 11 (0.352 + 0.002), PUA 1 (0.337 + 0.007), PUA 32 (0.328 + 0.02), PUA 6 (0.321 + 0.002) and PUA 16 (0.321 + 0.01) respectively. Further, the clear zone assay was performed using PEG as a carbon source, and the maximum zone of clearance was observed in isolates PUA 20, PUA 35, PUA 11, and PUA 6 after staining with Coomassie brilliant blue, which is shown in Figure 2.
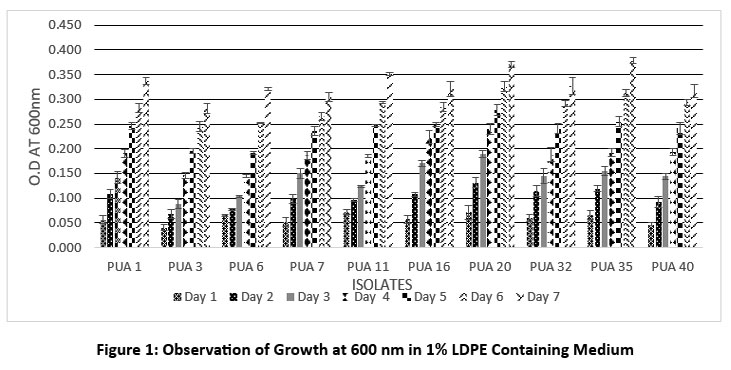 | Figure 1: Observation of Growth at 600 nm in 1% LDPE Containing Medium
|
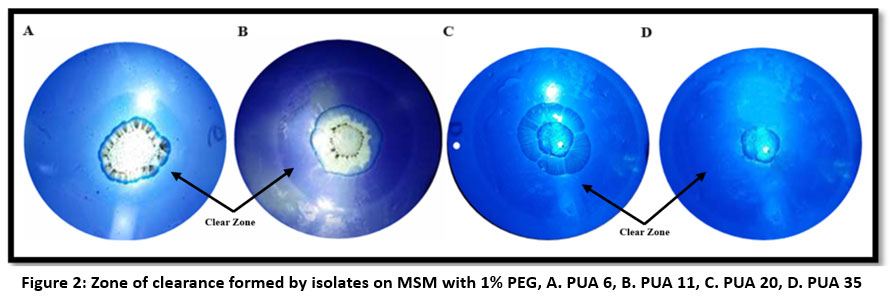 | Figure 2: Zone of clearance formed by isolates on MSM with 1% PEG, A. PUA 6, B. PUA 11, C. PUA 20, D. PUA 35
|
Screening of isolates for Hydrocarbon degradation
Hydrocarbon degradation is generally carried out by using a model alkane such as hexadecane, crude oil, or any other long-chain alkane. In this study n hexadecane was used as a model hydrocarbon. To screen out the hydrocarbon degrading potential of isolated actinomycetes (LDPE degrading) their growth was observed at 600nm in the presence of 1 % n hexadecane in MSM broth for 7 days at 120 rpm in shaking condition in 30 C. Two isolates namely PUA 11 and PUA 35 showed a highest growth in the initial stage (24 hour) compare to other isolates was 0.063 + 0.006 and 0.061 + 0.02 respectively (Figure 3). Upon completion of the incubation phase, the maximum growth was observed in PUA 35 (0.352 + 0.002), followed by PUA 20 (0.345 + 0.003), PUA 11 (0.336 + 0.002), and PUA 6 (0.310 + 0.002), respectively. In addition, to confirm the hexadecane degradation, the clear zone assay was carried out, and the isolates with the highest growth potential, such as PUA 35, PUA 20, PUA 11, and PUA 6, showed a zone of clearance on the MSM plate with hexadecane (Figure 4).
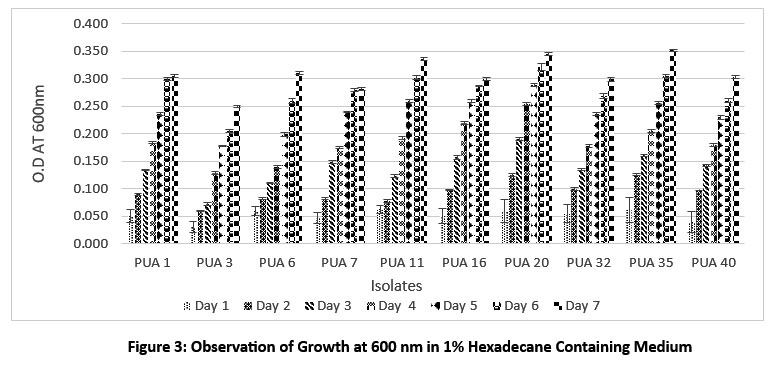 | Figure 3: Observation of Growth at 600 nm in 1% Hexadecane Containing Medium
|
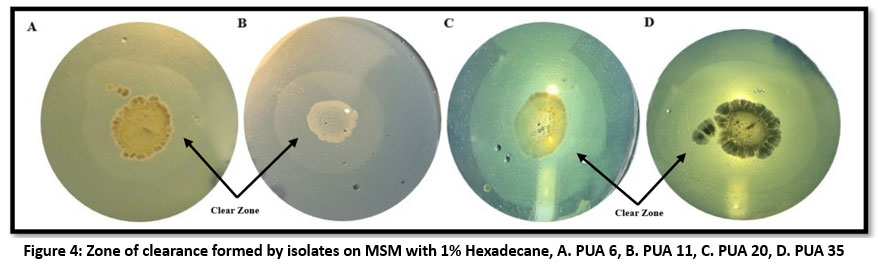 | Figure 4: Zone of clearance formed by isolates on MSM with 1% Hexadecane, A. PUA 6, B. PUA 11, C. PUA 20, D. PUA 35
|
Lab Scale Degradation LDPE Sheets
The actinomycete strains capable of decomposing LDPE powder were then tested for their capacity to degrade commercially available LDPE sheets, often used for packaging. To determine the level of degradation, a comparison was made between the control group (uninoculated LDPE sheets) and the test group (LDPE sheets inoculated with the isolates PUA 20, PUA 35, PUA 11, and PUA 6) after one month of incubation. The weight of the LDPE sheets in the inoculated samples was found to have significantly decreased compared to the control, indicating microbial degradation activity. The degradation performance of each potent isolate is presented in Figure 5. Among all the isolates, PUA 20 had the highest degrading efficiency, with a weight loss of 16.2% ± 0.2, followed by PUA 35 (15.5% ± 0.1), PUA 11 (14.6% ± 0.2), and PUA 6. These findings indicate that the selected actinomycete isolates have the potential to biodegrade LDPE materials.
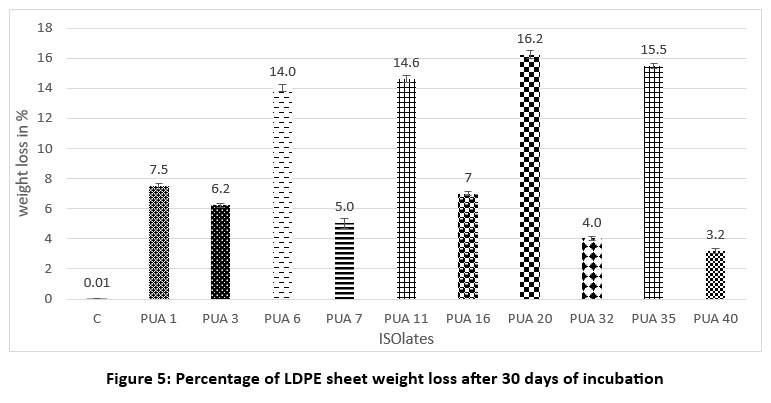 | Figure 5: Percentage of LDPE sheet weight loss after 30 days of incubation
|
FTIR analysis of LDPE Sheets
The FTIR spectroscopy was used to confirm that the chemical modification occurs on the LDPE sheets by the isolated actinomycetes. The FTIR spectrum of control sheet showed the absorbance peaks at 719.07 cm-1 (C-H rock), 1462.21 cm-1 (-C-H blending), and peaks at 2846.26 and 2913.35 cm-1, 2980.35 cm-1 indicates - C-H stretch (figure 6). which are typical for polyethylene and indicate the integrity of its aliphatic hydrocarbon backbone (Figure 6).
In comparison with the control, there were modifications in the absorbance peaks of the LDPE sheets inoculated with the isolates, which is shown in Figure 6. There was a presence of new absorbance peaks in PUA 20, PUA 35, and PUA 11, which were at 953.54 cm-1, 1073.79 cm-1, 1149.35 cm-1, and 1251.59 cm-1 (Figure 6). These additional peaks indicate the development of novel functional groups, including ether (C-O-C), carbonyl (C=O), and hydroxyl (-OH) moieties, indicating oxidative or hydrolytic breakdown of the polymer matrix. The appearance of these functional groups offers support to the theory that actinomycetes aided in the partial disintegration of the polyethylene structure by enzymatic activity, presumably involving oxidative enzymes such as laccases, peroxidases, or oxygenases. The observed changes in the FTIR spectra give molecular-level evidence of structural disruption and chemical transformation in the polymer chains, confirming the biodegradation capacity of the selected actinomycete isolates.
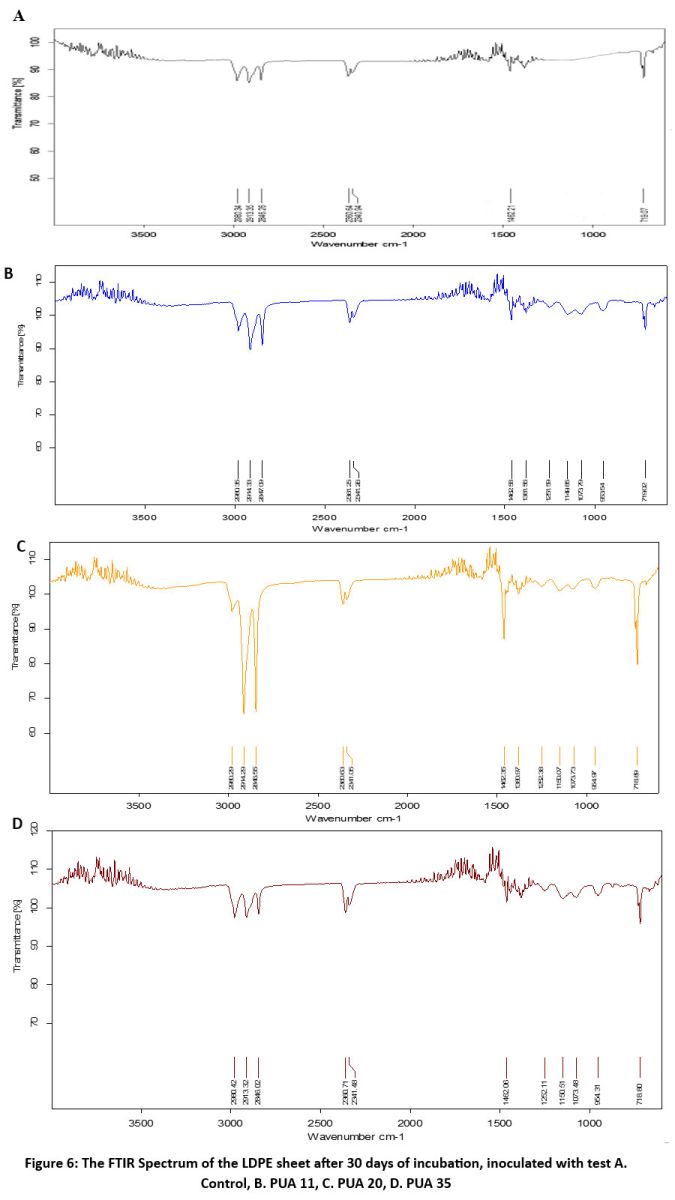 | Figure 6: The FTIR Spectrum of the LDPE sheet after 30 days of incubation, inoculated with test A. Control, B. PUA 11, C. PUA 20, D. PUA 35
|
Discussion
Plastic dumping sites or garbage dump sites are well studied for the understanding of plastic degradation mechanisms in earlier research and described as a major source of obtaining microbes with plastic-degrading abilities.
The lab-scale LDPE sheet degradation was confirmed by determination of weight loss after 1 month of incubation time. The weight loss determination is the simplest technique frequently used for the confirmation of LDPE degradation.
Conclusion
The present study emphasizes the promising capability of actinomycetes in the simultaneous biodegradation of low-density polyethylene (LDPE) and hydrocarbons, two major environmental pollutants. Among the 50 isolates screened from plastic dumping sites in Rajkot, Gujarat, several strains demonstrated notable efficacy in degrading LDPE sheets, with a maximum weight loss of 16.2% observed within a one-month incubation period. The ability of selected isolates to grow on hexadecane further confirmed their hydrocarbon-degrading capabilities. FTIR analysis provided strong evidence of polymer structural breakdown, revealing the formation of new functional groups indicative of LDPE degradation. These findings suggest that actinomycetes, with their versatile metabolic capabilities, hold significant promise as eco-friendly agents for bioremediation. Harnessing such microbial systems could contribute to integrated waste management strategies aimed at reducing plastic and hydrocarbon pollution, paving the way for more sustainable environmental practices.
Acknowledgement
We would like to thank the Department of Microbiology, Atmiya University for infrastructural support.
Funding Sources
The authors are grateful to the Gujarat government for financial support received under “SHODH SCHEME” (202201243).
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript includes all datasets generated or examined during this research investigation.
Ethics Statement
This study did not include human participants, animal subjects, or any material requiring ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Unnati Bhaveshbhai Yagnik: Conceptualization, Data Collection, Analysis, Writing original draft, Revision
Mousumi Bijoykumar Das: Supervision, Review, Revision
Reference
- Kumar Sen, S. & Raut, S. Microbial degradation of low density polyethylene (LDPE): A review. J Environ Chem Eng 3, 462–473 (2015).
CrossRef - Andrady, A. L. Microplastics in the marine environmentAndrady, A. L. (2011). Microplastics in the marine environment. Marine Pollution Bulletin, 62(8), 1596–1605. https://doi.org/10.1016/J.MARPOLBUL.2011.05.030. Mar Pollut Bull 62, (2011).
CrossRef - Geyer, R., Jambeck, J. R. & Law, K. L. Production, use, and fate of all plastics ever made. Sci Adv 3, 25–29 (2017).
CrossRef - Raddadi, N. & Fava, F. Biodegradation of oil-based plastics in the environment: Existing knowledge and needs of research and innovation. Science of the Total Environment vol. 679: 148-158https://doi.org/10.1016/j.scitotenv.2019.04.419 (2019).
CrossRef - Yip, J. & Chan, W. Y. Textile fibers and fabrics. Latest Material and Technological Developments for Activewear 47–72 (2020) doi:10.1016/B978-0-12-819492-8.00003-X.
CrossRef - India Plastics Pact. The India Plastics Pact Annual Report. (2022).
- Arpia, A. A., Chen, W.-H., Ubando, A. T., Naqvi, S. R. & Culaba, A. B. Microplastic degradation as a sustainable concurrent approach for producing biofuel and obliterating hazardous environmental effects: A state-of-the-art review. J Hazard Mater 418, 126381 (2021).
CrossRef - Busca, G. Production of gasolines and monocyclic aromatic hydrocarbons: From fossil raw materials to green processes. Energies (Basel) 14, (2021).
CrossRef - Pandolfo, E., Barra Caracciolo, A. & Rolando, L. Recent Advances in Bacterial Degradation of Hydrocarbons. Water (Switzerland) 15, (2023).
CrossRef - Kumar Shrestha, J., Raj Joshi, D., Regmi, P., Badahit, G. & Author, C. Isolation and Identification of Low Density Polyethylene (LDPE) Degrading Bacillus spp. from a Soil of Landfill Site Identification of functional marker for the gene coding brittleness trait in rice View project Isolation and Identification of Low Density. Acta Scientific Microbiology 2, 30–34 (2019).
- Soud, S. A. Biodegradation of polyethylene LDPE plastic waste using locally isolated Streptomyces sp. Journal of Pharmeceutical Sciences and Research 11, 1333–1339 (2019).
- Nowak, B., Pajak, J., Drozd-Bratkowicz, M. & Rymarz, G. Microorganisms participating in the biodegradation of modified polyethylene films in different soils under laboratory conditions. Int Biodeterior Biodegradation 65, 757–767 (2011).
CrossRef - Nanda, S. & Sahu, S. S. Biodegradability of polyethylene by Brevibacillus, Pseudomonas, and Rhodococcus spp. New York Science Journal 3, 95–98 (2010).
- Hadad, D., Geresh, S. & Sivan, A. Biodegradation of polyethylene by the thermophilic bacterium Brevibacillus borstelensis. J Appl Microbiol 98, 1093–1100 (2005).
CrossRef - Ndahebwa Muhonja, C., Magoma, G., Imbuga, M. & Makonde, H. M. Molecular characterization of Low-Density Polyethene (LDPE) degrading bacteria and fungi from Dandora dumpsite, Nairobi, Kenya. Int J Microbiol 2018, (2018).
CrossRef - Hussein, A. A., Al-mayaly, I. K. & Khudeir, S. H. Isolation , Screening and Identification of Low Density Polyethylene ( LDPE ) degrading bacteria from contaminated soil with plastic wastes. 1, 1–14 (2015).
- K.I.T, E., G.M., A. & O.A., O. Biodegradation of Used Engine Oil By Bacteria Isolated From Soil Contaminated With Used Engine Oil in Ogbomoso, Nigeria. IOSR J Environ Sci Toxicol Food Technol 8, 66–70 (2014).
CrossRef - Soumeya, S., Allaoueddine, B. & Hocine, A. K. Biodegradation of used motor oil by Streptomyces ginkgonis KM-1–2, isolated from soil polluted by waste oils in the region of Azzaba (Skikda-Algeria). J Biotechnol 349, 1–11 (2022).
CrossRef - Ahmed, R. S. & Swargiary, M. D. Plastic and Petroleum Hydrocarbon Degrading Potentials of Single and Mixed Bacterial Cultures Isolated from Garbage Areas of Darrang, Assam. Nature Environment and Pollution Technology 20, 275–280 (2021).
CrossRef - Yuan, Y. et al. Unique Raoultella species isolated from petroleum contaminated soil degrades polystyrene and polyethylene. Ecotoxicol Environ Saf 263, 115232 (2023).
CrossRef - Usha, R., Sangeetha, T. & Palaniswamy, M. Screening of polyethylene degrading microorganisms from garbage soil. Libyan Agriculture Research Center Journal International 2, 200–204 (2011).
- Auta, H. S., Emenike, C. U. & Fauziah, S. H. Screening of Bacillus strains isolated from mangrove ecosystems in Peninsular Malaysia for microplastic degradation. Environmental Pollution 231, 1552–1559 (2017).
CrossRef - Rana, K. & Rana, N. Isolation and Screening of Plastic Degrading Bacteria from Dumping Sites of Solid Waste. Int J Curr Microbiol Appl Sci 9, 2611–2618 (2020).
CrossRef - Denaro, R. et al. Marine hydrocarbon-degrading bacteria breakdown poly(ethylene terephthalate) (PET). Science of the Total Environment 749, (2020).
CrossRef - Kuswytasari, N. D. et al. Plastic Biodegradation Potential of Soil Mangrove Mold Isolated from Wonorejo, Indonesia. Adv Life Sci 10, 228–238 (2023).
- Dey, A. S., Bose, H., Mohapatra, B. & Sar, P. Biodegradation of Unpretreated Low-Density Polyethylene (LDPE) by Stenotrophomonas sp. and Achromobacter sp., Isolated From Waste Dumpsite and Drilling Fluid. Front Microbiol 11, 1–15 (2020).
CrossRef - Kumar, R. et al. Landfill microbiome harbour plastic degrading genes: A metagenomic study of solid waste dumping site of Gujarat, India. Science of the Total Environment 779, 146184 (2021).
CrossRef - Asmita, K., Shubhamsingh, T. & Tejashree, S. Isolation of plastic degrading micro-organisms from soil samples collected at various locations in Mumbai, India. International Research Journal of Environment Sciences 4, (2015).
- Nakei, M. D., Misinzo, G., Tindwa, H. & Semu, E. Degradation of polyethylene plastic bags and bottles using microorganisms isolated from soils of Morogoro, Tanzania. Front Microbiol 13, 1–15 (2022).
CrossRef - Soud, S. A. Biodegradation of polyethylene LDPE plastic waste using locally isolated Streptomyces sp. Journal of Pharmeceutical Sciences and Research 11, 1333–1339 (2019).
- Rana, K. & Rana, N. Isolation and Screening of Plastic Degrading Bacteria from Dumping Sites of Solid Waste. Int J Curr Microbiol Appl Sci 9, 2611–2618 (2020).
CrossRef - Ahmed, R. S. & Swargiary, M. D. Plastic and Petroleum Hydrocarbon Degrading Potentials of Single and Mixed Bacterial Cultures Isolated from Garbage Areas of Darrang, Assam. Nature Environment and Pollution Technology 20, 275–280 (2021).
CrossRef - Gajendiran, A., Krishnamoorthy, S. & Abraham, J. Microbial degradation of low-density polyethylene (LDPE) by Aspergillus clavatus strain JASK1 isolated from landfill soil. 3 Biotech 6, (2016).
CrossRef - R. Pramila. Biodegradation of low density polyethylene (LDPE) by fungi isolated from marine water– a SEM analysis. Afr J Microbiol Res 5, (2011).
CrossRef - Gajendiran, A., Krishnamoorthy, S. & Abraham, J. Microbial degradation of low-density polyethylene (LDPE) by Aspergillus clavatus strain JASK1 isolated from landfill soil. 3 Biotech 6, (2016).
CrossRef - Ibiene, A. A., Stanley, H. O. & Immanuel, O. M. Biodegradation of polyethylene by Bacillus sp. indigenous to the Niger Delta mangrove swamp. Niger J Biotechnol 26, 68–79 (2013).






